BP705P – Pharmaceutical Analysis Lab Manual
"Mastering Pharmaceutical Analysis: A Comprehensive Lab Manual for Aspiring Scientists"

Paid Advertisements

Free Courier service
Courier time: 8:00PM to 8:00 AM
-
BP406P – Medicinal Chemistry-I Lab ManualJanuary 1, 2025
-
BP701T- Unit-1 : Spectroscopic Basic and UV spectroscopyJuly 16, 2024
Printing Charges
- 65 paisa per printing page for back to back printing – Market rate Rs 1.5 to Rs 10
- 75 paisa per xerox page for back to back xerox – Market rate Rs 1.5 per side
- Rs 4* colour printing/xerox – Market rate Rs 5 to Rs 10
- Rs 1.5 per printing page for back to back printing on bonded paper Market rate Rs 5 to Rs 8
- Rs 6* colour printing/xerox on bonded paper – Market rate Rs 7 to Rs 10
- A3 Print/Xerox black and white – Rs 10 – Market rate Rs 15 to Rs 25
- A3 colour Print – Rs 25 – Market rate Rs 40 to Rs 90
- Lamination A4 – Rs 15 – Market rate Rs 20 to Rs 25
- Spiral Binding-Rs 20 Market rate Rs 35 to Rs 90
Table of Contents
Syllabus as per - "PCI"

- Determination of absorption maxima and effect of solvents on absorption
- Maxima of organic compounds
- Estimation of dextrose by colorimetry
- Estimation of sulfanilamide by colorimetry
- Simultaneous estimation of ibuprofen and paracetamol by UV spectroscopy
- Assay of paracetamol by UV- Spectrophotometry
- Estimation of quinine sulfate by fluorimetry
- Study of quenching of fluorescence
- Determination of sodium by flame photometry
- Determination of potassium by flame photometry
- Determination of chlorides and sulphates by nephelo turbidometry
- Separation of amino acids by paper chromatography
- Separation of sugars by thin layer chromatography
- Separation of plant pigments by column chromatography
- Demonstration experiment on HPLC
- Demonstration experiment on Gas Chromatography
Practical: 01: Calibration of UV Spectrophotometer
Blank page content
Aim
Calibration of the UV-Visible spectrophotometer
Observation table
Wavelength (nm) | Absorbance |
235 | 0.755 |
257 | 0.868 |
313 | 0.290 |
350 | 0.634 |
Calculations
Specific absorbance for 1%w/v solution

Result
The calibration of UV-Visible spectrophotometer was performed. Are parameters are performed and found to be in limit.
Line page content
Aim
Calibration of the UV-Visible spectrophotometer
Reference
Indian Pharmacopeia, 2014 (Volume-I), Indian Pharmacopeia commission ministry of health and Family welfare, Government of India, page no 139-141.
Requirement
Apparatus
Volumetric flask, Test tube, test tube stands, beaker, micropipette
Chemical
Potassium dichromate solution, sulphuric acid, distilled water
Instrument
UV Spectrophotometer, Weighing Balance
Theory
UV-visible spectrophotometry is used in the measurement of the absorption of monochromatic radiation by any substance solution in the range of 200 nm to 800 nm. The magnitude of absorbance is expressed in terms of absorbance (A) and is defined as the logarithm to base 10 of the reciprocal of transmittance (T).

Where,
I0 = Intensity of incident radiation
I = Intensity of transmitted radiation
According to Beer’s-Lambert’s law, the absorbance of a solution depends on the concentration of the absorbing substance in the solution and the thickness of the absorbing layer. For convenience and ease in calculations, the absorbance adopted in I.P for several substance is specific absorbance and is given by the formula
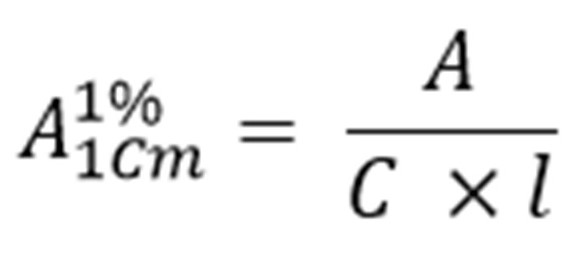
Where,
C = concentration of absorbing substance expressed in %w/v
l = Thickness of absorbing layer in cm
A = Absorbance
Preparation of reagent
0.005M Sulphuric acid
Pipette out 0.27 ml of concentrated sulphuric acid and transfer to 1000ml volumetric flask. To it add 800 ml of distilled water. Shake the flask and make up the volume up to mark with distilled water.
Potassium dichromate solution
Weigh 60 mg of potassium dichromate and transfer to 1000ml volumetric flask. To this flask add, 800 ml of 0.005M sulphuric acid. Dissolve the complete solid. After dissolution, make up the volume up to mark with 0.005M sulphuric acid.
Procedure
Control of absorbance
Check the absorbance by using suitable filters or a prepared solution of potassium dichromate. The table given below gives the idea for each wavelength and limits of specific absorbance.
Wavelength (nm) | Specific Absorbance | Maximum Absorbance |
235 | 124.5 | 122.9 to 126.2 |
257 | 144 | 142.8 to 145.7 |
313 | 148 | 47.0 to 50.3 |
350 | 106.6 | 104.9 to 108.2 |
430 | 15.9 | 15.7 to 16.1 |
Result
The calibration of UV-Visible spectrophotometer was performed. Are parameters are performed and found to be in limit.
Practical 2: Demonstration of UV Spectrophotometer
Blank page content
Aim
To understand the working, parts, and principle of a UV-Visible spectrophotometer through a live demonstration.
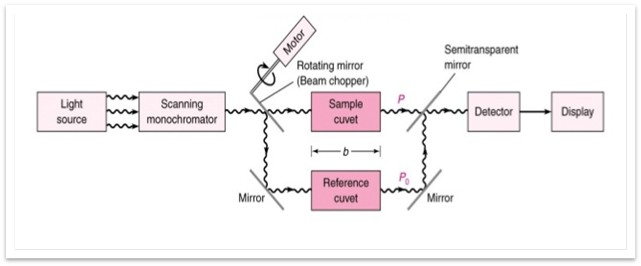
Figure of UV Spectrophotometer
Single Beam UV Spectrophotometer
Double Beam UV Spectrophotometer
UV Spectrum of Paracetamol
Result
The compound showed maximum absorbance at ___ nm.
The instrument correctly demonstrated the principle of absorbance measurement in UV-Vis spectroscopy.
Line page content
Aim
To understand the working, parts, and principle of a UV-Visible spectrophotometer through a live demonstration.
Reference
Backett A. H.; Stenlahe J. B. Practical pharmaceutical chemistry, 4th edition, part-II, CBS publisher, New delhi, page no 328
Requirement
Apparatus
Volumetric flask, Pipette, Beaker, test tube, funnel
Chemical
Paracetamol API, 0.1N NaOH Solution
Instrument
Weighing balance, UV spectrophotometer
Theory
A UV-Visible spectrophotometer is an analytical instrument used to measure the absorbance or transmittance of a substance in the ultraviolet (200–400 nm) and visible (400–800 nm) regions of the electromagnetic spectrum. The technique is based on Beer-Lambert’s law, which relates absorbance to concentration.
Beer-Lambert Law:
A = ε × c × l
Where:
A = Absorbance
ε = Molar absorptivity
c = Concentration of the solution
l = Path length of the cuvette (usually 1 cm)
When a substance absorbs UV or visible light, electrons are excited from ground state to higher energy levels. This absorbance is characteristic of the compound and can be used for quantitative and qualitative analysis.
Procedure
- Put the funnel on 10ml volumetric flask
- Transfer the ____ g (0.01g) of paracetamol to volumetric flask.
- Add 6ml of the prepared 0.1M sodium hydroxide solution slowly through funnel.
- Shake the content vigorously, so that all paracetamol gets dissolved.
- Make the volume with 0.1M sodium hydroxide and transfer 10ml to beaker.
- Transfer this solution to test tube and Label this solution as stock solution (SS)
- Pipette out 0.1 mL from SS and transfer this solution to 10 mL volumetric flask.
- Make the volume with 0.1M sodium hydroxide solution.
- Transfer the content to test tube and label this as D1 (dilution 1)
- Pipette out 0.1 mL from D1 and transfer this solution to 10 mL volumetric flask.
- Make the volume with 0.1M sodium hydroxide solution.
- Transfer the content to test tube and label this as D2 (dilution 2)
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting blank in both cuvette.
- Now put D2 or D1 solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Determine the wavelength where it shows highest absorbance.
Result
The compound showed maximum absorbance at ___ nm.
The instrument correctly demonstrated the principle of absorbance measurement in UV-Vis spectroscopy.
Practical 3: Effect of solvent
Blank page content
Aim
Study of effect of solvent on maximum absorbance of pyridine
Observations
Pyridine Spectrum in methanol
Pyridine Spectrum in distilled water
Overlay spectrum of pyridine in distilled water and methanol
Result
Pyridine shows maximum absorbance at ____ nm having absorbance of _____ in methanol whereas it shows maximum absorbance at ___ nm in water with the absorbance of ______ .
From the overlay spectra it is clear that there is not only change in max absorbance of pyridine but also change in change in absorbance.
Line page content
Aim
Study of effect of solvent on maximum absorbance of pyridine
Reference
Backett A. H.; Stenlahe J. B. Practical pharmaceutical chemistry, 4th edition, part-II, CBS publisher, New delhi, page no 328
Requirement
Apparatus
Density bottle, Volumetric flask, Micropipette, Test-tube, test-tube stand, tissue paper
Chemicals
Pyridine, Methanol and distilled water
Instruments
Weighing balance, UV-spectrophotometer
Theory
UV-spectroscopy, commonly known as ultraviolet-visible (UV-Vis) spectroscopy, is a popular analytical technique for determining the absorption of ultraviolet and visible light by molecules. The interaction of light with matter is the basic concept of UV-spectroscopy. The interaction of light with matter is based on electronic transition.
In organic molecules, there are three types of electrons.
- Sigma electrons (σ)
- Pie electrons (π)
- Non-bonded electrons (n).
Based on this, any organic compound can undergo four different types of electrical transitions.
- σ → σ*
- n → σ*
- π → π*
- n → π*
Type 1 transitions require the most energy, while type 4 transitions require the least energy (n → π*).
The wavelength and energy of light are inversely related. As a result, increasing the wavelength reduces the energy of light and vice versa.
Principle
The solvent in which a drug is dissolved might influence its maximum absorbance, often known as its maximum absorption wavelength (max). This is known as the “solvent effect” or “solvent shift.” When a drug is dissolved in different solvents, its absorbance spectra may alter, and the λ max may move to a new wavelength.
Several factors influence the solvent effect on a drug’s maximal absorbance:
Electronic transitions:
Light absorption by molecules happens as a result of electronic transitions between distinct energy levels. The polarity and electronic characteristics of the solvent can influence the specific electronic transitions that contribute to the absorbance spectrum of a medication.
Solvent polarity:
The polarity of the solvent can have a substantial impact on the maximum concentration of a medication. Polar solvents have the ability to stabilize charged species and change the energy levels of molecules, resulting in changes in the absorption spectra.
Hydrogen bonding:
The presence of hydrogen bonding interactions between the drug molecule and the solvent molecules can also affect the absorption spectra. Such interactions have the potential to change the distribution of electronic charge and the energy levels available for electronic transitions.
Solvent viscosity:
Viscous fluids can impede molecular movements and influence the relaxation of excited states, potentially altering the max.
Concentration and aggregation: Drugs may aggregate at high concentrations or in certain solvents, causing changes in the absorbance spectrum.
pH of the solvent:
pH of the solvent: For pharmaceuticals having ionizable functional groups, the pH of the solvent can affect the drug’s ionization state and, as a result, its absorbance spectrum.
Procedure
- Pipette 0.1mL of pyridine into a 10 mL volumetric flask.
- Add around 5 mL of methanol.
- Shake the flask vigorously until a homogenous solution forms.
- Increase the volume by adding methanol until the desired volume is reached.
- Pour this solution into a test tube and label it methanolic stock solution.
- If required, dilute further with methanolic stock solution.
- Repeat steps 1–5 with water instead of methanol.
- Using UV spectroscopy, scan resulting methanolic and aqueous solutions from 200 nm to 400 nm and determine the individual maximum absorbance.
- Observe for change in λ max of pyridine in both solvents
Result
Pyridine shows maximum absorbance at ____ nm having absorbance of _____ in methanol whereas it shows maximum absorbance at ___ nm in water with the absorbance of ______ .
From the overlay spectra it is clear that there is not only change in max absorbance of pyridine but also change in change in absorbance.
Practical 4: Effect of pH
Blank page content
Aim
To study the effect of pH on the λmax of sulphanilamide.
Observations

Spectrums of sulphanilamide
Spectrum of sulphanilamide in Methanol
Spectrum of sulphanilamide in 0.1N Methanol HCl
Spectrum of sulphanilamide in 0.1N Methanol NaOH
Overlay Spectrum of sulphanilamide in Methanol, 0.1N Methanol HCl and 0.1N Methanol NaOH
Result
From the overlay spectrum it is observed that the spectrum obtained in methanol and basic pH there is no alteration in auxochrome and chromophore of sulfanilamide but in acidic pH there is significant alteration in auxochrome. Due to this there is change in λmax as well as change in absorbance.
Line page content
Aim
To study the effect of pH on the λmax of sulphanilamide.
Reference
Backett A. H.; Stenlahe J. B. Practical pharmaceutical chemistry, 4th edition, part-II, CBS publisher, New delhi, page no 328
Requirement
Apparatus
Beaker, Volumetric flask, Pipette, measuring cylinder, funnel, test tube, test tube stand.
Chemical
Sulphanilamide, hydrochloric acid, sodium hydroxide, distilled water
Instrument
Weighing balance, UV-Visible spectrophotometer
Theory
λmax term is used in UV-Visible spectroscopy where one looks at electronic transitions between the HOMO level and the LUMO level. Molecules absorb light over a range of wavelengths and when this absorbance is plotted as a function of wavelength one obtains an absorption spectrum. An absorption spectrum is simply a plot of absorbance vs wavelength. This plot gives us a range of wavelengths within which a molecule absorbs.
Lambda max refers to the wavelength in the absorption spectrum where the absorbance is maximum. Generally, molecules absorb in a wavelength range centered around the lambda max. It acts as a single quantitative parameter to compare the absorption range of different molecules.
Sometimes spectral shifts occur due to changes in solvent polarity, changes in molecular structure etc. All such spectral shifts (i.e., blue shift or red shift) can be quantified with shift in lambda max. For example, increase in conjugation length in a molecule shifts the lambda max to the red.
The wavelength at which a substance has its strongest photon absorption. It gives the maximum probable value of the wavelength of the absorbance of UV radiation by the sample under investigation. it is also important in UV spectroscopy because it gives an accurate value of wavelength for a specific substance.
Principal
Sulphanilamide is basic in nature. The terminal amino group has non-bonded lone pair of electrons. This make them auxochrome. In basic pH there is no disturbance in non-bonded electrons means the auxochromic effect is present. In acidic pH the terminal amino group loose non-bonded electron due to salt formation. This means that at acidic pH, auxochromic effect of sulphanilamide will be lost and spectrum will be at different wavelength with different absorbance.
Procedure
- Put the funnel on 10ml volumetric flask
- Transfer the 10 mg (10μg/ml) of sulphanilamide to volumetric flask.
- Add 6ml of methanol and mix the content. Share vigorously till all sulphanilamide dissolve.
- Make the volume with methanol.
- Transfer this solution to the test tube and label it as stock solution.
- Pipette out 0.1ml from this stock solution and transfer it to 10 ml volumetric flask.
- Make the volume the with methanol and label it as D1 (means Dilution 1)
- Pipette out 0.1 ml from D1 and transfer to volumetric flask. Make the volume with methanol and label it as D2 (means dilution 2).
- Pipette out 0.1 ml from D1 and transfer to volumetric flask. Make the volume with 0.1N HCl and label it as D2 Acidic (means dilution 2 in acidic pH).
- Pipette out 0.1 ml from D1 and transfer to volumetric flask. Make the volume with 0.1N NaOH and label it as D2 nasic (means dilution 2 basic).
- Prepare the blank solution by performing all steps but without adding sulphanilamide. Means there will be two blank one for acidic solution and one for basic solution.
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting acidic blank solution in both cuvette.
- Now put acidic solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Determine the wavelength and absorbance.
- Repeat the step 13 to 15 for basic solution and methanolic sulphanilamide solution.
Result
From the overlay spectrum it is observed that the spectrum obtained in methanol and basic pH there is no alteration in auxochrome and chromophore of sulfanilamide but in acidic pH there is significant alteration in auxochrome. Due to this there is change in λmax as well as change in absorbance.
Practical 5: Determination of Max. Absorbance
Blank page content
Aim
To determine the λmax of paracetamol API
Observation
Observations
- Weight of weighing bottle + paracetamol : ________ g
- Weight of weighing bottle after transfer : ________ g
- Weight of paracetamol taken (1-2) (say A) : ________ g/___ ml
- Reported λmax of paracetamol : 257 nm
Spectrum
Paracetamol
Overlay spectrum
Dilution Tree
For paracetamol

Result
The λmax of paracetamol and nimesulide in a 0.1 M sodium hydroxide solution was found to be ____ nm and ____ nm respectively.
Line page content
Aim
To determine the λmax of paracetamol API
Reference
Watson David G. Pharmaceutical Analysis – A text book for pharmacy students and pharmaceutical chemists. 2nd edition, elesevier publication, page number 108-110.
Requirement
Apparatus
Beaker, Volumetric flask, Pipette, measuring cylinder, funnel, test tube, test tube stand.
Chemicals
Sodium hydroxide pellets, distilled water
Instruments
Weighing balance, UV-Visible spectrophotometer
Theory
λmax term is used in UV-Visible spectroscopy where one looks at electronic transitions between the HOMO level and the LUMO level. Molecules absorb light over a range of wavelengths and when this absorbance is plotted as a function of wavelength one obtains an absorption spectrum. An absorption spectrum is simply a plot of absorbance vs wavelength. This plot gives us a range of wavelengths within which a molecule absorbs.
Lambda max refers to the wavelength in the absorption spectrum where the absorbance is maximum. Generally, molecules absorb in a wavelength range centered around the lambda max. It acts as a single quantitative parameter to compare the absorption range of different molecules.
Sometimes spectral shifts occur due to changes in solvent polarity, changes in molecular structure etc. All such spectral shifts (i.e., blue shift or red shift) can be quantified with shift in lambda max. For example, increase in conjugation length in a molecule shifts the lambda max to the red.
The wavelength at which a substance has its strongest photon absorption. It gives the maximum probable value of the wavelength of the absorbance of UV radiation by the sample under investigation. it is also important in UV spectroscopy because it gives an accurate value of wavelength for a specific substance.
Preparation of reagent/Solvent
In a clean dry 100 mL volumetric flask, transfer 0.4g of sodium hydroxide. To it add approximately 50 mL of freshly boiled and cooled distill water. Shake the content of flask till all sodium hydroxide gets dissolved. After complete dissolution of sodium hydroxide, make the volume with fresh boiled and cooled distill water. The resulting solution will have the strength of 0.1M/0.1N NaOH.
Procedure
- Put the funnel on 10ml volumetric flask
- Transfer the ____ g (0.01g) of paracetamol to volumetric flask.
- Add 6ml of the prepared 0.1M sodium hydroxide solution slowly through funnel.
- Shake the content vigorously, so that all paracetamol gets dissolved.
- Make the volume with 0.1M sodium hydroxide and transfer 10ml to beaker.
- Transfer this solution to test tube and Label this solution as stock solution (SS)
- Pipette out 0.1 mL from SS and transfer this solution to 10 mL volumetric flask.
- Make the volume with 0.1M sodium hydroxide solution.
- Transfer the content to test tube and label this as D1 (dilution 1)
- Pipette out 0.1 mL from D1 and transfer this solution to 10 mL volumetric flask.
- Make the volume with 0.1M sodium hydroxide solution.
- Transfer the content to test tube and label this as D2 (dilution 2)
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting blank in both cuvette.
- Now put D2 or D1 solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Determine the wavelength where it shows highest absorbance.
Result
The λmax of paracetamol and nimesulide in a 0.1 M sodium hydroxide solution was found to be ____ nm and ____ nm respectively.
Practical No 6: linearity curve of paracetamol in 0.1M NaOH solution
Blank page content
Aim
To construct the linearity curve of paracetamol in 0.1M NaOH solution.
Observations
- Weight of weighing bottle + paracetamol API : ________ g
- Weight of weighing bottle after transfer : ________ g
- Weight of paracetamol API taken (1-2) (say A) : ________ g
- λmax chosen for building the linearity curve : 257nm
- Concentration of Paracetamol API in stock solution: _______ ug/ml
- Concentration of Paracetamol API in working stock solution: _______ ug/ml
Overlay spectrum
Observation table
Sr. | Volume of solution | Concentration of | Absorbance |
|
|
|
|
Excel graph
Result
The linearity curve of paracetamol in 0.1M NaOH solution was successful/ unsuccessful constructed with the r2 Value of _______ and linear equation of _______.
Line page content
Aim
To construct the linearity curve of paracetamol in 0.1M NaOH solution.
Reference
Backett A. H.; Stenlahe J. B. Practical pharmaceutical chemistry, 4th edition, part-II, CBS publisher, New delhi, page no 328-335
Requirement
Appartus
Volumetric flask, Test tube, test tube stands, beaker, micropipette
Chemical
Paracetamol API, sodium hydroxide, distilled water
Instruments
Weighing balance, UV-visible spectrophotometer
Theory
λmax term is used in UV-Visible spectroscopy where one looks at electronic transitions between the HOMO level and the LUMO level. Molecules absorb light over a range of wavelengths and when this absorbance is plotted as a function of wavelength one obtains an absorption spectrum. An absorption spectrum is simply a plot of absorbance vs wavelength. This plot gives us a range of wavelengths within which a molecule absorbs.
Lambda max refers to the wavelength in the absorption spectrum where the absorbance is maximum. Generally, molecules absorb in a wavelength range centered around the lambda max. It acts as a single quantitative parameter to compare the absorption range of different molecules.
Sometimes spectral shifts occur due to changes in solvent polarity, changes in molecular structure etc. All such spectral shifts (i.e., blue shift or red shift) can be quantified with shift in lambda max. For example, increase in conjugation length in a molecule shifts the lambda max to the red.
The wavelength at which a substance has its strongest photon absorption. It gives the maximum probable value of the wavelength of the absorbance of UV radiation by the sample under investigation. it is also important in UV spectroscopy because it gives an accurate value of wavelength for a specific substance.
Principle
The principle involved in UV estimation of paracetamol is based on the absorption of UV light by the molecule. The absorption of UV light is based on the presence of chromophore.
A linearity curve, also known as a calibration curve or standard curve, is a graphical representation of the relationship between the concentration of a substance (analyte) and its absorbance . The linearity curve is used to quantify the amount of analyte in an unknown sample by comparing its measured response to the responses of standard solutions with known concentrations.
The linearity curve serves as a tool for quantification, allowing you to convert the measured response (absorbance) of an unknown sample into a corresponding concentration value. A well-constructed linearity curve demonstrates the instrument’s sensitivity, precision, and accuracy in measuring the analyte’s concentration. It’s important to note that the linearity of the curve indicates the range over which the method is accurate and linear. Outside this range, the instrument’s response may deviate from linearity, and dilutions or adjustments to the method might be necessary.
Preparation of reagents
0.1 M Sodium Hydroxide
In a clean dry 100 mL volumetric flask, transfer 0.4g of sodium hydroxide. To it add approximately 50 mL of freshly boiled and cooled distill water. Shake the content of flask till all sodium hydroxide gets dissolved. After complete dissolution of sodium hydroxide, make the volume with fresh boiled and cooled distill water. The resulting solution will have the strength of 0.1M/0.1N NaOH.
Procedure
- Put the funnel on 10 mL volumetric flask
- Transfer the ____ g of paracetamol API to volumetric flask.
- Add approximate 6 mL of the prepared 0.1M sodium hydroxide solution slowly through funnel.
- Shake the content vigorously, so that all paracetamol gets dissolved.
- Make the volume with 0.1M sodium hydroxide label this solution as stock solution.
- From this stock solution, _____ ml was pipette out and was transfer to ___ ml volumetric flask. Add the 0.1M solution to make the volume. Label this solution as working stock solution.
- From the stock solution 1, pipette out 1 ml, 2 ml, 3ml, 4 ml, 5 ml, 6 ml, 7 ml, 8 ml, 9 ml, and 10 ml. Transfer this pipetted amount to 10ml volumetric flask.
- Make the volume with 0.1M NaOH solution
- Use 0.1M NaOH solution as blank.
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting blank in both cuvettes.
- Now put measuring solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Measure the absorbance of paracetamol at 257 nm.
- Repeat step 12 and 13 for other working solution.
- On the graph paper plot the graph of concetration versus absorbance and observed the graph.
- If maximum point touches the line then the linearity curve is successfully constructed.
Result
The linearity curve of paracetamol in 0.1M NaOH solution was successful/ unsuccessful constructed with the r2 Value of _______ and linear equation of _______.
Practical No 7: Assay of Paracetamol
Blank page content
Aim
To perform the assay of paracetamol tablet as per I.P.
Observations
- Weight of twenty tablets (Say A) : ________ g
- Average weight of one table (Say B) : ________ g
- Label claim (Say C) : ________ mg
- Weight of weighing bottle + paracetamol powder : ________ g
- Weight of weighing bottle after transfer : ________ g
- Weight of paracetamol powder taken (4-5) (say D): ________ g
- Absorbance of paracetamol powder (say E) : ________
- Molar absorptivity value of paracetamol (say F) : 715
- Volume of original extract (say G) : 20 ml
Spectrum of paracetamol tablet
Calculations
Expected content in tablet powder taken (Say H)
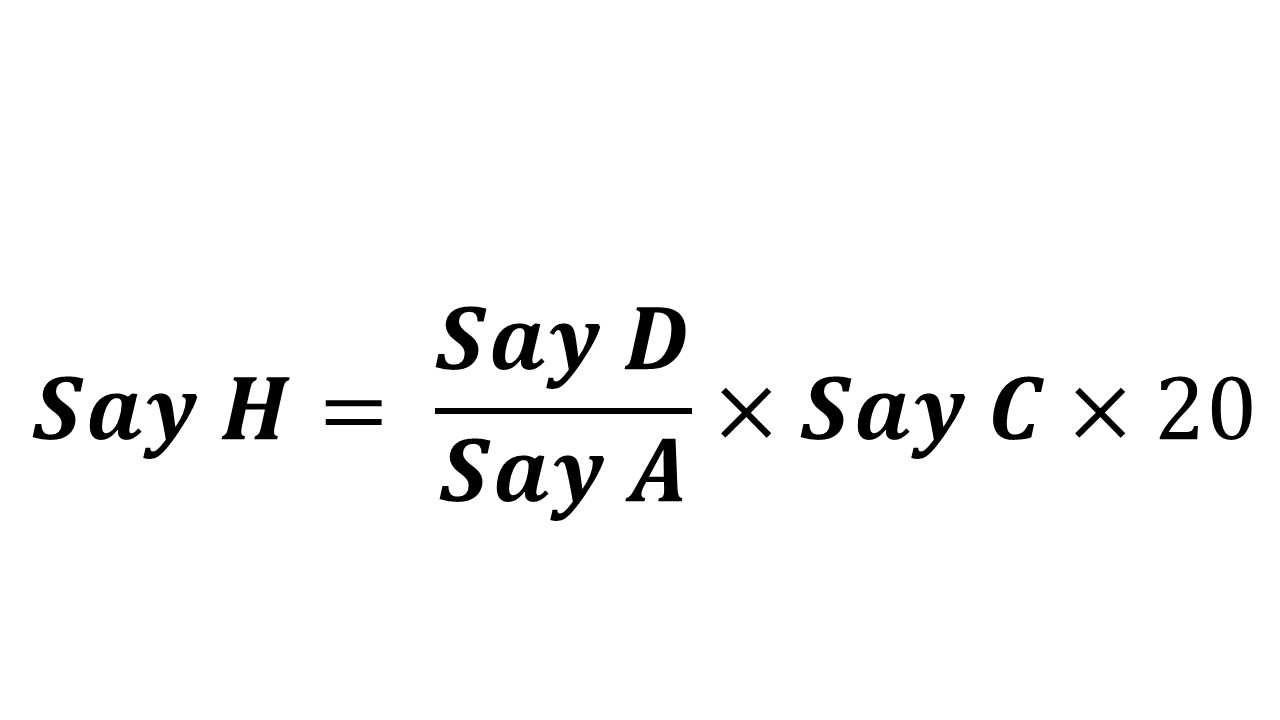
= ____ mg (Say I)
Dilution factor
1st Dilution = 1ml → 10 ml = 10
2nd Dilution = 1ml → 10 ml = 10
Total Dilution factor = 10 X 10 = 100 (Say J)
Concentration of paracetamol in diluted tablet extract (Say K)

= ____ g/100ml (Say K)
= _____ mg/100 ml (Say L)
Concertation of paracetamol in original extract solution
= Say L X Say J
= _____ mg/100ml (Say M)
100ml contains Say M paracetamol
Say G ml contains –

= ____ mg (Say N)
Percentage purity of paracetamol in given tablet is
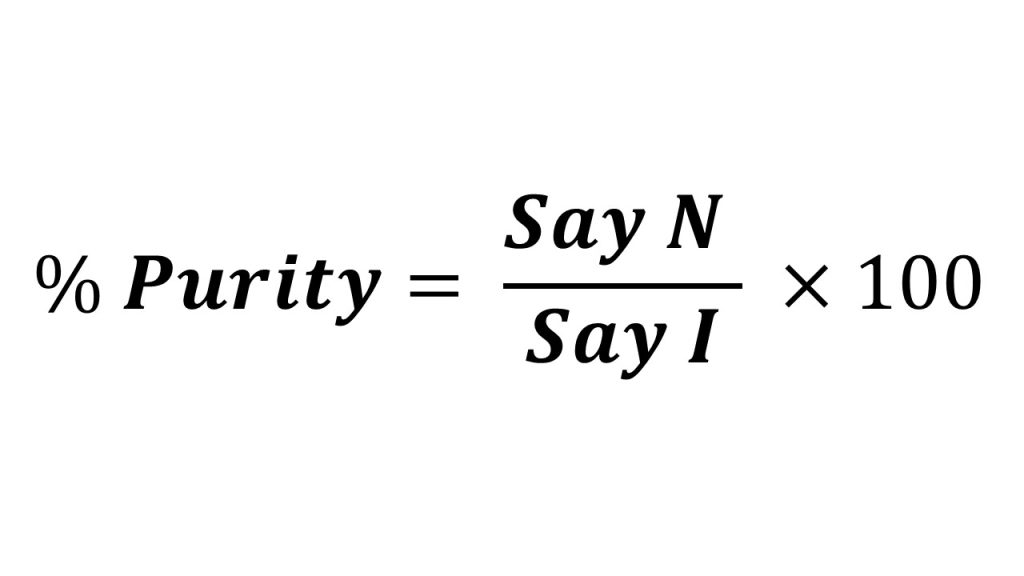
Result
The % purity of paracetamol in the given tablet formulation was found to be _____, and according to I. P., the given tablet passed/failed for content.
Line page content
Aim
To perform the assay of paracetamol tablet as per I.P.
Reference
Indian Pharmacopeia, 2014 (Volume-I), Indian Pharmacopeia commission ministry of health and Family welfare, Government of India, page no 139-141.
Requirements
Apparatus
Volumetric flask, Test tube, test tube stands, beaker, micropipette
Chemicals
Paracetamol tablet, sodium hydroxide, distilled water
Instrument
Weighing balance, UV-visible spectrophotometer
Theory
λmax term is used in UV-Visible spectroscopy where one looks at electronic transitions between the HOMO level and the LUMO level. Molecules absorb light over a range of wavelengths and when this absorbance is plotted as a function of wavelength one obtains an absorption spectrum. An absorption spectrum is simply a plot of absorbance vs wavelength. This plot gives us a range of wavelengths within which a molecule absorbs.
Lambda max refers to the wavelength in the absorption spectrum where the absorbance is maximum. Generally, molecules absorb in a wavelength range centered around the lambda max. It acts as a single quantitative parameter to compare the absorption range of different molecules.
Sometimes spectral shifts occur due to changes in solvent polarity, changes in molecular structure etc. All such spectral shifts (i.e., blue shift or red shift) can be quantified with shift in lambda max. For example, increase in conjugation length in a molecule shifts the lambda max to the red.
The wavelength at which a substance has its strongest photon absorption. It gives the maximum probable value of the wavelength of the absorbance of UV radiation by the sample under investigation. it is also important in UV spectroscopy because it gives an accurate value of wavelength for a specific substance.
Principle
The assay of paracetamol is based on interaction of π electrons with UV radiations. The solubility of paracetamol is poor in water, to increase the solubility and to extract paracetamol from tablet powder, sodium hydroxide solution is used. The terminal phenolic hydroxyl will react with strong base to form sodium phenoxide compound which is easily soluble in water. The rest of tablet residue then easily be filter. Also water and sodium hydroxide solution is transparent to UV.
Preparation of 0.1M Sodium hydroxide solution
In a clean dry 100 mL volumetric flask, transfer 0.4g of sodium hydroxide. To it add approximately 50 mL of freshly boiled and cooled distill water. Shake the content of flask till all sodium hydroxide gets dissolved. After complete dissolution of sodium hydroxide, make the volume with fresh boiled and cooled distill water. The resulting solution will have the strength of 0.1M/0.1N NaOH.
Procedure
- Put the funnel on 10ml volumetric flask
- Transfer the ____ g of paracetamol powder to volumetric flask.
- Add 6ml of the prepared 0.1M sodium hydroxide solution slowly through funnel.
- Shake the content vigorously, so that all paracetamol gets dissolved.
- Make the volume with 0.1M sodium hydroxide and filter the content. transfer the filter 10ml to volumetric flask and again make the volume if required. Transfer this content to beaker.
- Measure more 10ml 0.1M sodium hydroxide solution by volumetric flask and transfer this solution to beaker.
- Mix the content and label the content as stock solution
- Pipette out 1 ml of stock solution and transfer it to 10ml volumetric flask.
- Make the volume of this solution with 0.1M NaOH solution and label it as stock solution 1
- Pipette out 1ml from stock solution and transfer it to 10ml of volumetric flask. Make the volume with 0.1M NaOH solution and label it as final dilution
- Prepare the blank solution by performing all steps but without adding paracetamol
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting blank in both cuvette.
- Now put measuring solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Measure the absorbance of paracetamol at 257 nm.
- Determine the content and percent purity at the maximum at 257 nm taking 715 as specific absorbance of paracetamol at 257 nm.
- Determine the wavelength where it shows highest absorbance.
Result
The % purity of paracetamol in the given tablet formulation was found to be _____, and according to I. P., the given tablet passed/failed for content.
Practical No 8: Concentration of paracetamol vby Linear graph method, linear equation method, single point standarization, and Double point standarization
Blank page content
Aim
To determine the unknown concentration of paracetamol by the linear graph method, linear equation method, single point standardization, and double point standardization method.
Observations
- Weight of twenty tablets (Say A) : ________ g
- Average weight of one table (Say B) : ________ g
- Label claim (Say C) : ________ mg
- Weight of weighing bottle + paracetamol powder : ________ g
- Weight of weighing bottle after transfer : ________ g
- Weight of paracetamol powder taken (4-5) (say D): ________ g
- Absorbance of paracetamol powder (Test) (say E) : ________
- Absorbance of paracetamol standard (S1) : ________
- Absorbance of paracetamol standard (S2) : ________
- Volume of original extract (say F) : 20 ml.
Observation table
Sr. No | Method | Result (ug/mL) |
1 | Linear graph method | ___ (Say G1) |
2 | Liner equation method | ___ (Say G2) |
3 | Single point standardization | ___ (Say G3) |
4 | Double point standardization | ___ (Say G4) |
Calculations
Expected content in tablet powder taken (Say H)
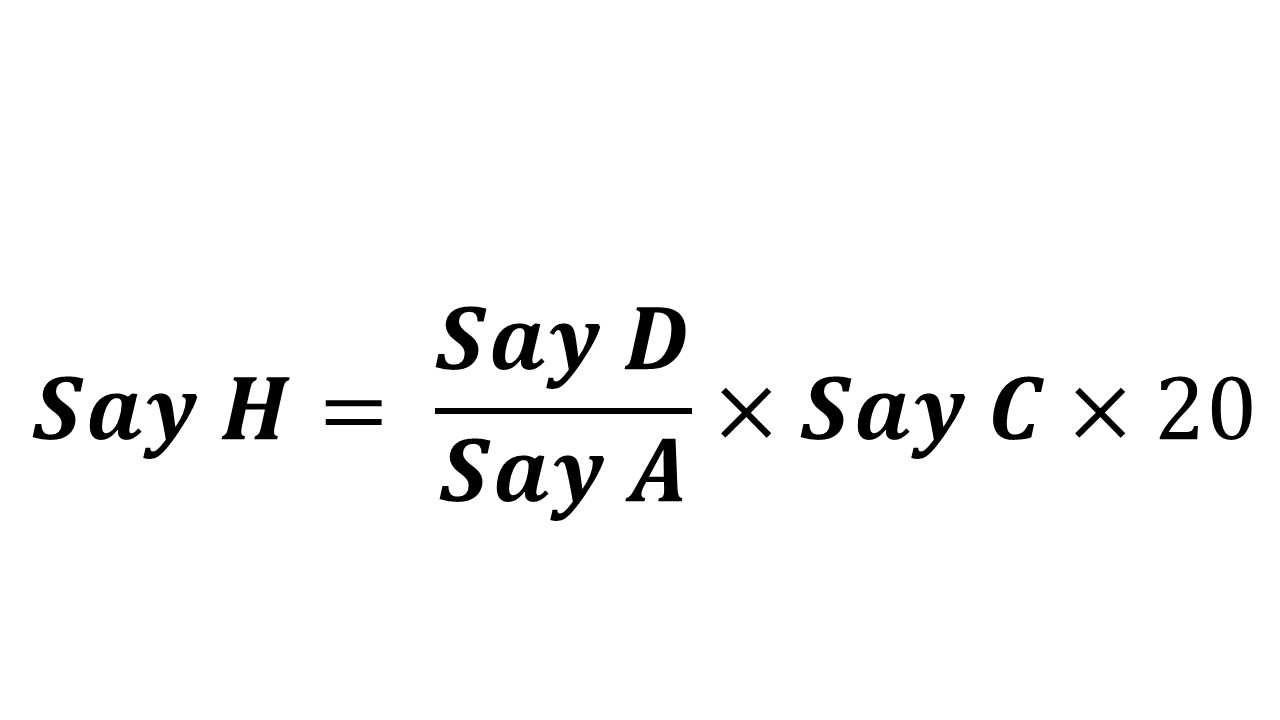
= ____ mg (Say I)
Dilution factor
5ml → 100 ml = 20 (Say J)
Concentration of paracetamol in diluted tablet extract determined by different method
Concentration of paracetamol by linear graph method
After extrapolation of absorbance from Y-axis to linearity curve and then extra plotting to x-axis i.e. concentration axis, the concentration of paracetamol obtained is _________ (SayG1)
Converting G1 ug/ml to mg/100ml
1ml → G1
100ml → G1*100
= ____ ug/100ml (Say K1)
Dividing K1 ug by 1000 gives _____ mg/100ml (Say L1)
Concentration of paracetamol in original extract
= Say L1 × Say J
= ______ mg/100ml (Say M1)
100ml contains SayM1 diclofenac
Say F ml contains
= (Say M1 × Say F)/100
= Say N1
Percentage purity of paracetamol in given tablet is
% purity = (Say N1 × 100)/Say I
= Say O1 %
Concentration of Paracetamol by Linear equation method
After plotting the excel graph and adding tredline, we receive the equation. Putting the value of absorbance in equation, we get the concentration of paracetamol in dilute extract, which is found to be ___ (Say G2) ug/ml
Converting G2 ug/ml to mg/100ml
1ml → G2
100ml → G2*100
= ____ ug/100ml (Say K2)
Dividing K2 ug by 1000 gives _____ mg/100ml (Say L2)
Concentration of paracetamol in original extract
= Say L2 × Say J
= ______ mg/100ml (Say M2)
100ml contains SayM1 paracetamol
Say F ml contains
= (Say M2 × Say F)/100
= Say N2
Percentage purity of paracetamol in given tablet is
% purity = (Say N2 × 100)/Say I
= Say O2 %
Concentration of paracetamol by single point standardization
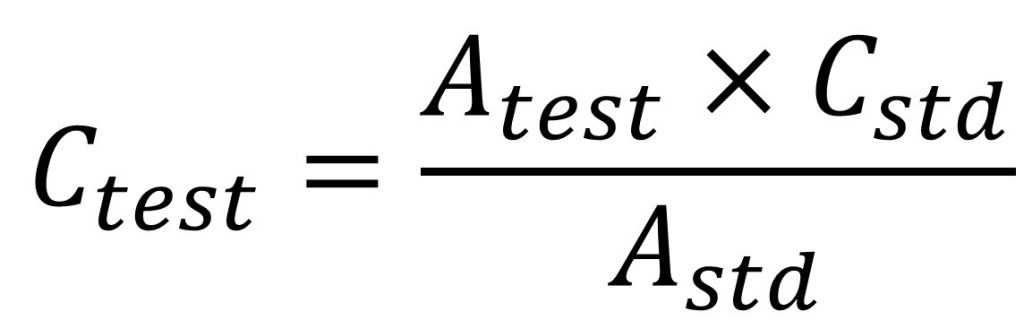
Putting the value of absorbance of test obtain, absorbance and concentration of standard used, the concentration of paracetamol in diluted solution was found to be ____ (Say G3) ug/ml
Converting G3 ug/ml to mg/100ml
1ml → G3
100ml → G3*100
= ____ ug/100ml (Say K3)
Dividing K3 ug by 1000 gives _____ mg/100ml (Say L3)
Concentration of paracetamol in original extract
= Say L3 × Say J
= ______ mg/100ml (Say M3)
100ml contains SayM3 paracetamol
Say F ml contains
= (Say M3 × Say F)/100
= Say N3
Percentage purity of paracetamol in given tablet is
% purity = (Say N3 × 100)/Say I
= Say O3 %
Concentration of paracetamol by double point standardization method
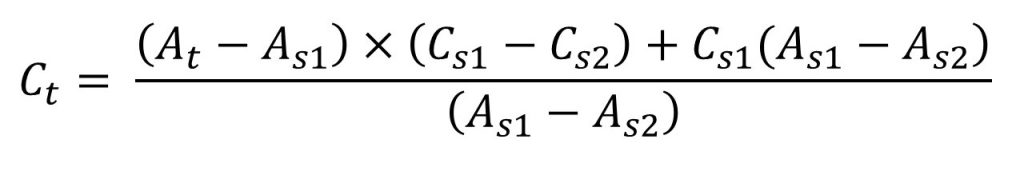
Putting the value of absorbance of test obtained, absorbance and concentration of two standard used, the concentration of paracetamol in diluted solution was found to be ____ (Say G4) ug/ml
Converting G4 ug/ml to mg/100ml
1ml → G4
100ml → G4*100
= ____ ug/100ml (Say K4)
Dividing K4 ug by 1000 gives _____ mg/100ml (Say L4)
Concentration of paracetamol in original extract
= Say L4 × Say J
= ______ mg/100ml (Say M4)
100ml contains SayM3 paracetamol
Say F ml contains
= (Say M4 × Say F)/100
= Say N4
Percentage purity of paracetamol in given tablet is
% purity = (Say N4 × 100)/Say I
= Say O4 %
Result
The % purity of paracetamol in given tablet formulation was found to be –
- Linear graph method: Say O1
- Linear equation method: Say O2
- Single point standardization: Say O3
- Double point standardization: Say O4
From this observation, we can say that there is minute difference in concentration so any one method can be used to determine the concentration of paracetamol in given tablet extract
According to I. P. the given tablet passes/fails for content.
Line page content
Aim
To determine the unknown concentration of paracetamol by the linear graph method, linear equation method, single point standardization, and double point standardization method.
Reference
Indian Pharmacopeia, 2007 (Volume-I), Indian Pharmacopeia commission ministry of health and Family welfare, Government of India, page no 139-141.
Requirements
Apparatus
Volumetric flask, Test tube, test tube stands, beaker, micropipette
Chemicals
Paracetamol tablet, sodium hydroxide, distilled water
Instrument
Weighing balance, UV-visible spectrophotometer
Theory
Determination of diclofenac in any formulation can be done by using –
- Calibration graph method
In this method, the number of standard solutions were prepared using reference substance and their absorbance were recorded. After getting the absorbance the graph was plotted by taking concentration on x-axis and absorbance on y-axis. The linear graph was then constructed passing from origin. Now, measure the absorbance of test analyte solution and put on the linear curve and from that determine the concentration of unknown solution.
- Linear equation method
In this method first the linear curve was constructed. After constructing the linear curve, the equation for straight line was obtain i.e. Y = mx + C.
Where,
Y → Absorbance of unkwown solution,
m → slope of line,
x → concentration of unknown solution,
C → Y or X intercept.
Putting the value of absorbance of unknown solution in this equation, directly the concentration of test solution can be determined.
- By using standard absorptivity value
This method was widely used by various pharmacopeia. This method is applicable for the stable analyte that are remains unaffected by variation in instrumental parameters like scanning speed, slit width etc. The use of standard absorptivity values prevents the preparation of standard solution of the reference standard substance for determination of absorptivity. It is also advantageous where the reference standard is difficult to obtain, or it is more expensive. The concentration of analyte is calculated by using following formula.
A = ε × c × b
Where,
A → Absorbance of test analyte,
ε→ Molar absorptivity,
c → Concentration of test analyte,
b → cell path length
- Using single point standardization method
Single point standardization method involves the measurement of the absorbance of a sample solution and standard solution. The sample solution and standard solution are prepared in identical manner. The absorbance of standard and test solution is determined. From the list of standard absorbance, one absorbance is selected which has absorbance close to the absorbance of test solution. Now the concentration of test solution is determined by using following formula
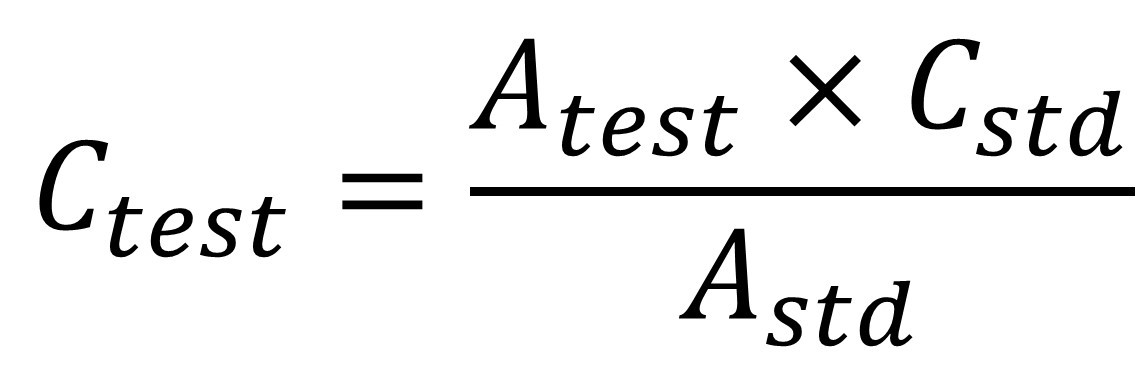
Where,
Ctest → Concentration of test solution,
Cstd → Concentration of standard solution,
Atest → Absorbance of test solution,
Astd → Absorbance of standard solution.
- By using double point standardization method
In the single point standard, accuracy is limited. To overcome this problem multiple point standardization method is used. In the method series of standard solution were prepared. In the identical manner the test solution was also prepared. The absorbance of all solution was recorded. From the absorbance of standard solutions, two absorbance were selected. The selection was done with respect to the test solution absorbance. The first standard absorbance that was selected has the absorbance less than test absorbance. The second standard absorbance which was selected has the absorbance more than standard solution. The concentration of test solution was determined from the formula as-
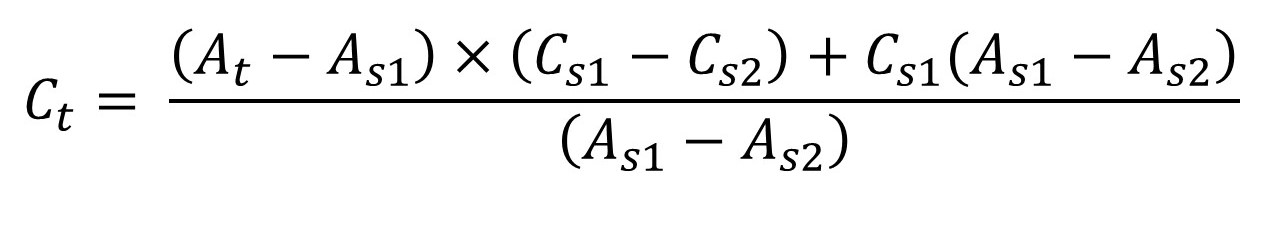
Where,
Ctest → Concentration of test solution,
Cstd → Concentration of standard solution,
Atest → Absorbance of test solution,
Astd → Absorbance of standard solution.
Principle
The assay of paracetamol is based on interaction of π electrons with UV radiations.
Preparation of 0.1M Sodium hydroxide solution
In a clean dry 100 mL volumetric flask, transfer 0.4g of sodium hydroxide. To it add approximately 50 mL of freshly boiled and cooled distill water. Shake the content of flask till all sodium hydroxide gets dissolved. After complete dissolution of sodium hydroxide, make the volume with fresh boiled and cooled distill water. The resulting solution will have the strength of 0.1M/0.1N NaOH.
Procedure
- Put the funnel on 10ml volumetric flask
- Transfer the ____ g of paracetamol powder to volumetric flask.
- Add 6ml of the prepared 0.1M sodium hydroxide solution slowly through funnel.
- Shake the content vigorously, so that all paracetamol gets dissolved.
- Make the volume with 0.1M sodium hydroxide and filter the content. transfer the filter 10ml to volumetric flask and again make the volume if required. Transfer this content to beaker.
- Measure more 10ml 0.1M sodium hydroxide solution by volumetric flask and transfer this solution to beaker.
- Mix the content and label the content as stock solution
- Pipette out 1 ml of stock solution and transfer it to 10ml volumetric flask.
- Make the volume of this solution with 0.1M NaOH solution and label it as stock solution 1
- Pipette out 1ml from stock solution and transfer it to 10ml of volumetric flask. Make the volume with 0.1M NaOH solution and label it as final dilution
- Prepare the blank solution by performing all steps but without adding paracetamol
- Now, start the UV and allow it run all checks.
- Perform the base line corrections by putting blank in both cuvette.
- Now put measuring solution in one cuvette and blank in another cuvette and scan the solution from 400 nm to 200 nm.
- Measure the absorbance of paracetamol at 257 nm.
- Determine the content and percent purity at the maximum at 257 nm taking 715 as specific absorbance of paracetamol at 257 nm.
- Determine the wavelength where it shows highest absorbance.
Result
The % purity of paracetamol in given tablet formulation was found to be –
- Linear graph method: Say O1
- Linear equation method: Say O2
- Single point standardization: Say O3
- Double point standardization: Say O4
From this observation, we can say that there is minute difference in concentration so any one method can be used to determine the concentration of paracetamol in given tablet extract
According to I. P. the given tablet passes/fails for content.
Blank page content
Practical No 9
Aim
To perform the ascending paper chromatography for given known amino acid
Observations
- Distance travelled by solute A : ________ cm
- Distance travelled by solute B : ________ cm
- Distance travelled by solute A and B in mixture : ____ and _____ cm
- Distance travelled by solvent front : ________ cm
Paper chromatogram

Calculations

Result
The Rf value of solute A and solute B was found to be ______ and _____ respectively
Line page content
Aim
To perform the ascending paper chromatography for given known amino acid
Reference
Chatwal G. R; Anand S. K. Instrumental method of chemical analysis, 5th edition, Himalaya Publication House, page number 2.588-2.590
Requirements
Apparatus
Developing chamber, Saturation paper, Whatman filter paper, micropipette, sprayer
Chemicals
Amino acid sample, Ninhydrin solution, Mobile phase
Theory
Chromatography is a physicochemical method for separation of complex mixtures and was discovered in 1903 by Russian-Italian botanist M. S. Tswett. Chromatography is a modern and sensitive technique used for rapid and efficient analysis and separation of components of a mixture and separation of components of a mixture and purification of compounds.
Basis of principle of chromatographic technique is based on the differential migration of the individual components of a mixture through a stationary phase under the influence of moving phase.
Based on stationary phase, the differential migration occurs.
Principle
The principle involved in paper chromatography is based on partition chromatography. The solute gets partition between the organic phase and aqueous phase. The latter is present in the filter paper. Thus the rf value of solute defines the affinity of solute towards mobile phase. If the rf value of solute is 1 means solute gets partition in mobile phase and if the rf value is 0 means solute gets partition to stationary phase.
There are different types paper chromatography
- Ascending paper chromatography
- Descending paper chromatography
- Ascending-descending paper chromatography
- 2D paper chromatography
- Radial chromatography
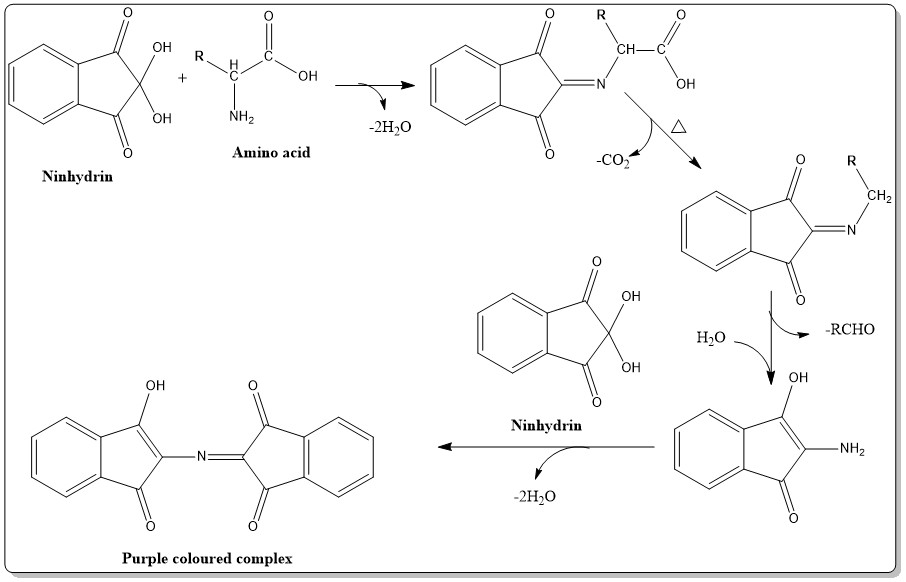
Reaction of amino acid with ninhydrin
Procedure
Preparation of paper
Cut the paper towel: 2 cm by 15 cm. Then, measure 4 cm from one end and draw a line using a pencil and also a midline dividing the 4 cm line. This is the baseline. It’s important to use pencil because lead will not dissolve in water, preventing experimental error in chemical separation. After this draw a small dot at the cross section of two lines you have drawn previously. Just opposite side of baseline of paper, bend the paper and insert the thread. After this stapple the paper so that thread will not move out.
Preparation of mobile phase
In the 100ml volumetric flask, add 70 ml of isopropyl alcohol. The volume was made up to mark with methanol.
Preparation of solute solution
The given amino acids were dissolved in suitable solvent.
Preparation of chamber
Pour the mobile phase that has been prepared and put the filter paper in that. Apply the lid and saturate the chamber for 30 minutes. While saturation, do not disturb the chamber.
Development of paper
At the mark point, apply the solute A solution, evaporate the solvent by using hot air dryer. Reapply the solute A spot. Repeat the same procedure by for solute B spot and for mixture spot.
Put the paper slowly in the chamber and apply the lid. Make sure, while developing the paper, chamber did not get disturb.
Result
The Rf value of solute A and solute B was found to be ______ and _____ respectively
Calculations
To perform the ascending paper chromatography for given unknown amino acid
In the 100ml volumetric flask, add 70 ml of isopropyl alcohol. The volume was made up to mark with methanol.
Practical No 10
Blank page content
Aim
To perform the radial paper chromatography for given known amino acid
Observations
- Distance travelled by solute A : ________ cm
- Distance travelled by solute B : ________ cm
- Distance travelled by solute A and B in mixture: _____ and ____ cm
- Distance travelled by solvent front : ________ cm
Paper
Calculations

Result
The Rf value of solute A and solute B was found to be ______ and _____ respectively.
Line page content
Aim
To perform the radial paper chromatography for given known amino acid
Reference
Chatwal G. R; Anand S. K. Instrumental method of chemical analysis, 5th edition, Himalaya Publication House, page number 2.588-2.590
Requirements
Apparatus
Developing chamber, Saturation paper, Whatman filter paper, micropipette, sprayer, petri-dish
Chemicals
Amino acid sample, Ninhydrin solution
Theory
Chromatography is a physicochemical method for separation of complex mixtures and was discovered in 1903 by Russian-Italian botanist M. S. Tswett. Chromatography is a modern and sensitive technique used for rapid and efficient analysis and separation of components of a mixture and separation of components of a mixture and purification of compounds.
Basis of principle of chromatographic technique is based on the differential migration of the individual components of a mixture through a stationary phase under the influence of moving phase.
Based on stationary phase, the differential migration occurs.
Principle
The principle involved in paper chromatography is based on partition chromatography. The solute gets partition between the organic phase and aqueous phase. The latter is present in the filter paper. Thus the rf value of solute defines the affinity of solute towards mobile phase. If the rf value of solute is 1 means solute gets partition in mobile phase and if the rf value is 0 means solute gets partition to stationary phase.
There are different types paper chromatography
- Ascending paper chromatography
- Descending paper chromatography
- Ascending-descending paper chromatography
- 2D paper chromatography
- Radial chromatography
Reaction of amino acid with Ninhydrin solution

Procedure
Take a circle Whatman filter paper and mark the centre by drawing line. Make a hole with a pin at this point. Make a wick of cotton and insert it through the hole of the chromatogram. In a petridish, place the solvent mixture and cover it with another petridish. Remove the wick from the chromatogram and apply the given mixture of amino acid using a micro-pipette to spot at the center of chromatogram. Blow the hot air to remove the solvent and repeat the spotting two times and insert the wick back when the spot has dried. Introduce the system of chromatogram and wick into the chromatography chamber (containing the solvent system). Ensure that the chromatogram rests on the rim of the petridish and does not dip into it. The chromatogram should remain in a stretched state and now cover it with another petridish. Leave the system undisturbed to allow the development of chromatogram. When the solvent front has almost reached three-fourth of the chromatogram, remove it and immediately mark the solvent front with a pencil. Spray the chromatogram with spraying reagent (Ninhydrin reagent) to visualize the separated components. Calculate Rf value of each components.
Result
The Rf value of solute A and solute B was found to be ______ and _____ respectively.
Practical No 11
Blank page content
Aim
Demonstration of working of fluorimeter
Figure
Observation table
Sr. No. | Concentration (μg/mL) | Fluorescence intensity |
1 |
|
|
2 |
|
|
3 |
|
|
4 |
|
|
5 |
|
|
6 |
|
|
7 |
|
|
8 |
|
|
9 |
|
|
Result
- Emission maximum observed at around 450 nm (for quinine sulfate).
- Fluorescence intensity increases with concentration up to a certain limit.
- Working of fluorimeter successfully demonstrated.
Line page content
Aim
Demonstration of working of fluorimeter
Reference
- Lakowicz, J. R. Principles of Fluorescence Spectroscopy, 3rd Edition, Springer, 2006.
- Skoog, D. A., Holler, F. J., Crouch, S. R. Principles of Instrumental Analysis, 6th Edition, Cengage Learning, 2007.
- Chatwal, G. R., Anand, S. Instrumental Methods of Chemical Analysis, 5th Edition, Himalaya Publishing House, 2010.
- Willard, H. H., Merritt, L. L., Dean, J. A., Settle, F. A. Instrumental Methods of Analysis, 7th Edition, CBS Publishers, 1986.
- Vogel, A. I. Vogel’s Textbook of Practical Organic Chemistry, 5th Edition, Pearson, 1996.
Requirements
Apparatus
Quartz cuvettes, Micropipette, Volumetric flasks, Test tube stand, Test tube, Funnel, Beaker
Chemicals
Fluorescent dye (e.g., quinine sulfate in 0.05 M H₂SO₄ / fluorescein in water), distilled water, solvent blank.
Instruments
Fluorimeter, Weighing Balance
Theory
Fluorescence is the emission of light by a substance that has absorbed light of shorter wavelength. The fluorimeter uses a monochromatic excitation source (usually UV light), and the emitted light at longer wavelength (Stokes shift) is detected at 90° to avoid interference from incident light. The intensity of fluorescence is proportional to the concentration of the fluorescent species within the linear range.
Principle
When a solution of a fluorescent compound is irradiated with UV light, electrons are excited to higher singlet states. On returning to the ground state, energy is released as fluorescence at a longer wavelength. The fluorimeter measures this emission and provides both qualitative (spectra) and quantitative (intensity vs. concentration) information.
Procedure
- Switch on the fluorimeter and allow it to stabilize.
- Place a quartz cuvette with solvent (blank) in the sample holder; set baseline to zero.
- Prepare dilute solutions of fluorescent standard (e.g., quinine sulfate).
- Excite the sample at a selected wavelength (e.g., ~350 nm for quinine sulfate).
- Record the emission spectrum (typically 360–500 nm).
- Compare intensity of emission at peak wavelength for different concentrations.
- Demonstrate linear relationship between concentration and fluorescence intensity (within low concentration range).
Result
- Emission maximum observed at around 450 nm (for quinine sulfate).
- Fluorescence intensity increases with concentration up to a certain limit.
- Working of fluorimeter successfully demonstrated.
.....
.....
A
A
.....
……….