BP701T- Unit-1 : Spectroscopic Basic and UV spectroscopy
"Simplifying Spectroscopy: A Comprehensive Guide to the Basics"

Table of Contents
Basics of spectroscopy
Introduction
Spectroscopy is the study of how light (electromagnetic radiation) interacts with matter in all forms. The interaction might produce electronic excitations (e.g., UV), molecular vibrations (e.g., IR), or nuclear spin orientations (e.g., NMR).
Spectroscopy is the most essential and promising method for studying the structural features of chemically coupled molecules. It is concerned with the interaction of electromagnetic radiation with matter and has the potential to extract incredibly important information such as the structure and other physicochemical properties of molecules. The oscillations of electric and magnetic dipoles in the atom generate electromagnetic radiations. The most important consequence of electromagnetic interaction is that matter absorbs or emits energy in discrete amounts known as quanta. Spectroscopic techniques are routinely employed to identify atomic and molecular structures, as well as the energy difference between different molecular energy states.
-
BP705P – Pharmaceutical Analysis Lab ManualJuly 12, 2024
Various spectroscopy techniques and quantum chemistry methods can provide useful information on molecular structure.
Electronic spectra
Electronic spectra result from transitions between electronic energy levels. This happens in the visible or ultraviolet bands of the electromagnetic spectrum. It contains information about molecular orbitals and bonding.


Vibrational transitions
Vibrational transitions occur in the infrared region of the electromagnetic spectrum and reveal information about organic molecules’ functional groups and bonds. The energy required to accomplish such transitions is less than that needed to construct electronic spectra.
Radiofrequencies
Radiofrequencies reveal information on the chemical environment of hydrogen atoms, as well as the number of hydrogen and carbon atoms in organic compounds.
These several fields of spectroscopy are mainly concerned with two experimental parameters:
- The energy of radiation absorbed or released by molecules.
- Spectral line intensity
The findings are connected to the electrical and molecular structures of the substances.
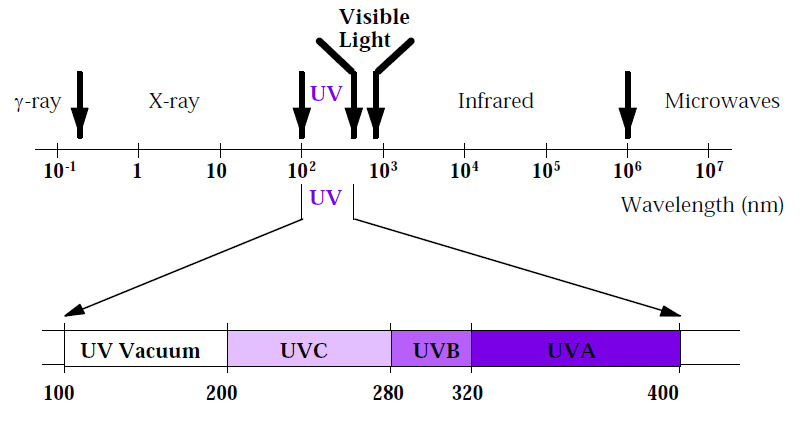
Electromagnetic Spectrum
The electromagnetic spectrum encompasses all electromagnetic radiation frequencies, wavelengths, and photon energies. The electromagnetic spectrum includes radio waves with the longest wavelengths and gamma rays with the shortest wavelengths. Spectroscopy may be used to investigate and describe materials using almost any type of electromagnetic radiation.

Some essential definitions
Electromagnetic radiations
Electromagnetic radiation is the transmission of electromagnetic waves/photons over space, conveying electromagnetic radiant energy. Electromagnetic waves are synchronized oscillations of electric and magnetic fields in vacuum that move at the speed of light. The oscillations of the two fields produce a transverse wave since they are perpendicular to each other as well as the direction of energy and wave propagation. Radio waves, microwaves, infrared radiations, visible light, ultraviolet radiations, X-rays, and gamma rays are all forms of electromagnetic radiation.
When charged particles are accelerated, electromagnetic waves are produced, which can then interact with other charged particles. Waves carry energy, momentum, and angular momentum away from their source particle, which can be transmitted to the matter with which they interact. Photons are electromagnetic wave quanta having a mass of zero.

Wavelength (λ)
The wavelength is defined as the space between any two successive portions of a wave whose vibrations are in phase.
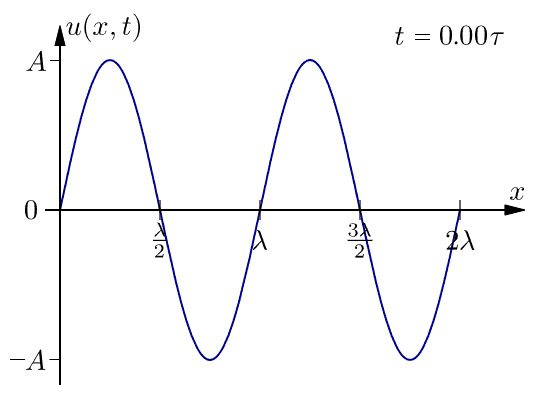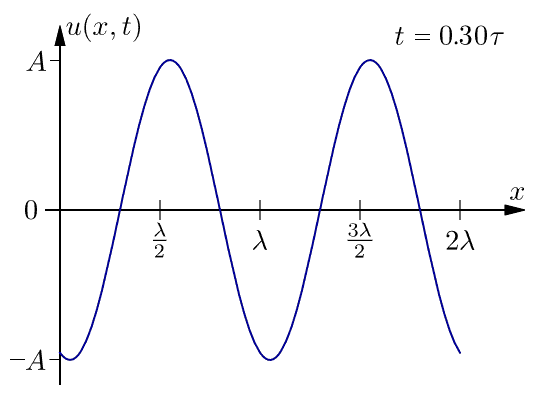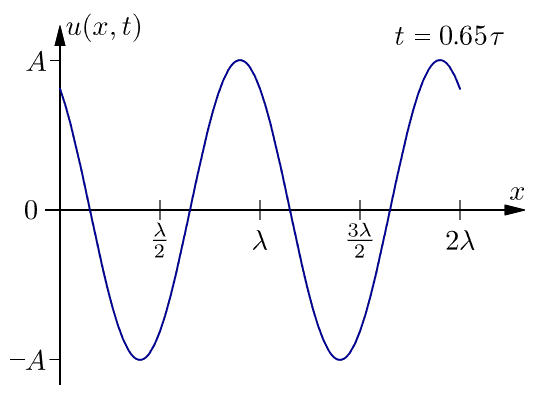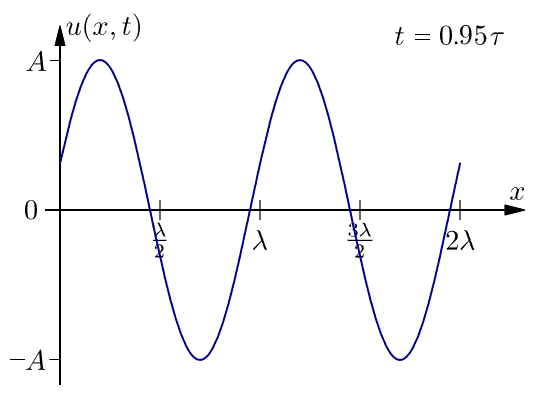
Frequency (v)
The number of waves crossing a spot in one second is referred to as its frequency.
Wave number
Wave number is defined as the number of wavelengths per unit distance.
Velocity (c)
Velocity of light is dependent on the medium through which it is passing and is defined as product of wavelength and frequency.
Relations of wavelength, frequency, vavenumber and energy

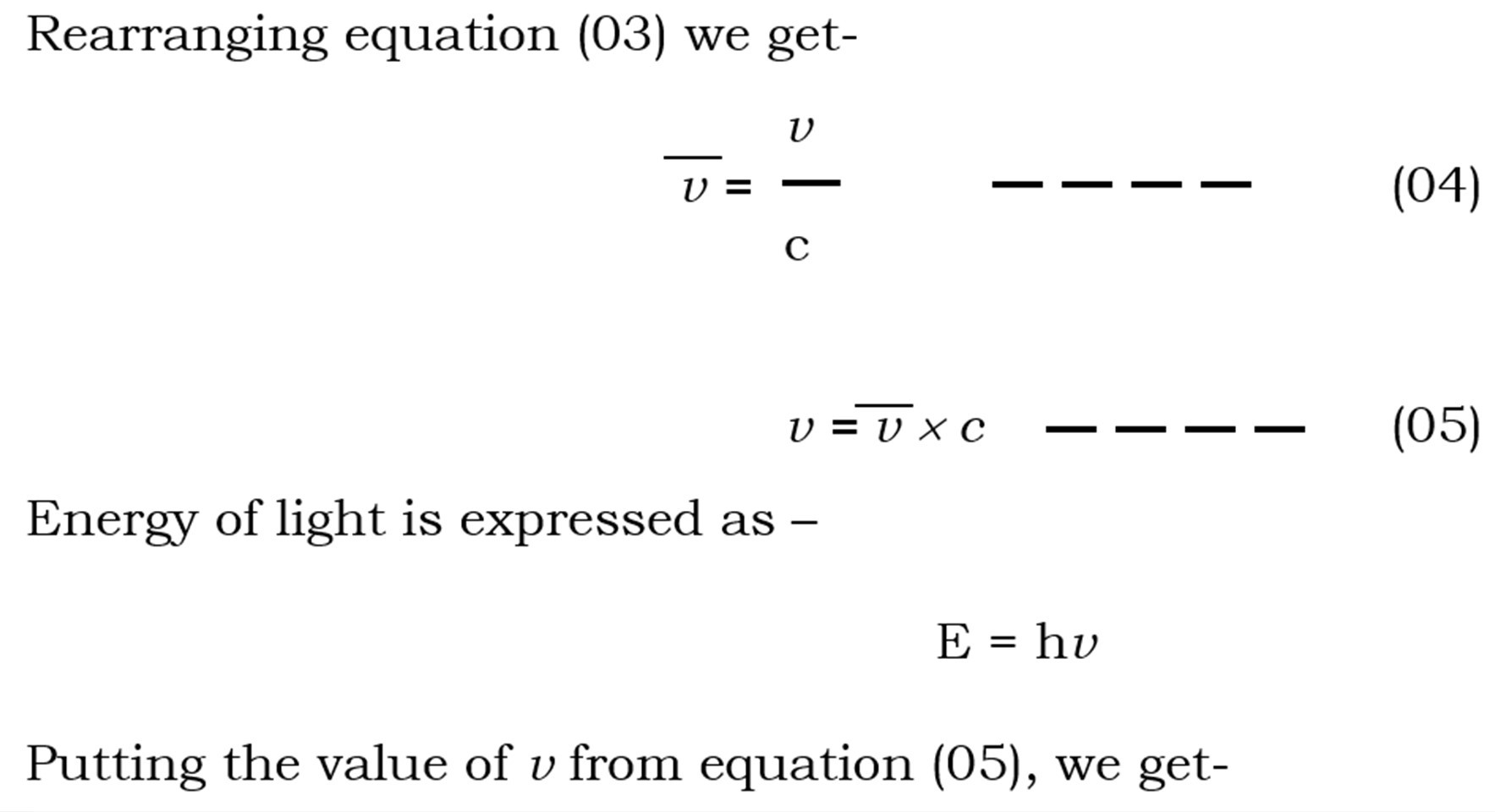

Absorbance
Absorbance is defined as log I0/It, which is equal to the reciprocal of the common logarithm of transmission.

Transmittance
Transmittance is defined as the ratio of log It/Io.

Photometer
A photometer is a device that measures light intensity or the optical properties of solutions or surfaces. It controls brightness, illumination, absorption, scattering, reflection, fluorescence, and phosphorescence.
A photometer is a device that measures the radiant power ratio of two electromagnetic beams across a defined wavelength range.
Spectrophotometer
A spectrophotometer is a device that measures the radiant power ratio of two electromagnetic beams across a wide wavelength range.
A spectrophotometer is a scientific apparatus that detects the intensity of light at various wavelengths. It has applications in a range of fields, including chemistry, physics, biology, and environmental science. A spectrophotometer’s principal function is to determine how much light a sample absorbs or transmits as a function of wavelength.
The spectrophotometer is based on the idea that various substances absorb distinct wavelengths of light. A sample may absorb certain wavelengths of light while transmitting others as it passes through it.
The detector measures the intensity of light before and after it passes through the sample, and the difference between the two intensities represents the sample’s absorbance at a certain wavelength.
Colorimeter
A colorimeter is any device that measures absorption in the visual range.
Classification of spectroscopy
Spectroscopy has been classified in several ways. The categorization system is based on-
- Types of radiative energy
- Nature of interaction
- Type of material
Types of radiative energy
- Electromagnetic radiation methods are classified by wavelength area, including microwave, infrared, near infrared, visible, ultraviolet, x-ray, and gamma spectroscopy.
- Electrons and neutrons can create radiative energy. Kinetic energy determines a particle’s wavelength.
Nature of interaction
The nature of the interaction between energy and matter can help distinguish between various forms of spectroscopy.
- Interactions may include absorption, which occurs when a substance absorbs radiative radiation.
- The term “emission” refers to a material’s ability to emit radiation.
- Elastic scattering and reflection spectroscopy measure how a material reflects or scatters incoming light.
- Impedance spectroscopy measures a medium’s capacity to impede energy transfer.
- Inelastic scattering occurs when radiation and matter exchange energy, causing the wavelength to shift. There are two examples: Raman and Compton scattering.
- In coherence spectroscopy, the radiative energy creates a coherent interaction between two quantum states of a material, which is sustained by the radiation field.
Type of material
Spectroscopic research are meant to observe how radiant energy interacts with various forms of substances.
- Atoms
- Molecule
- Nuclei
- Crystals and extended materials
Atomic spectra
Electrons follow a defined orbit around the nucleus, which comprises protons and neutrons. Orbital energy potential varies. Ionization energy is the amount of energy required to excite an electron out of its normal form. The electron is moved from its assigned point in the orbit by absorbing energy from an external source. The energy it absorbs to attain the excited state is returned to the atom when it returns to its original position. The radiated energy is measured with a spectrometer. A spectrum is a collection of energy levels in the form of radiation emitted by an excited atom. Each atom creates a discontinuous line spectrum. Each spectra line corresponds to a distinct wavelength and is unique to each element. In their spectra, no two elements have the same line pattern. The atomic spectrum refers to the radiation emitted by an atom.
The term “atom spectra” describes “those lines graphed on a spectroscope of specific wavelengths emitted by an electron as it returns from an excited state to its normal state.” In general, they are the atom’s outermost and least tightly bound electrons.”
Molecular spectra
A molecular spectrum is a radiation spectrum produced by electron transitions and other quantum energy changes within molecules, consisting of a series of unique spectral bands.
In contrast to atomic spectra, which are made up of tiny resonance lines, molecular spectra are distinguished by the absorption or emission of light across a substantially greater wavelength range. This is due to the fact that molecules may undergo a large number of transitions, while atoms can only undergo a few electrical changes.
Electromagnetic radiation of a certain wavelength that is absorbed by molecules can increase
- Electron energy is transferred to higher energy states, comparable to atomic absorption and UV-visible spectroscopy theory.
- Increased inter-nuclear vibrational energy of component atoms (IR Spectroscopy Principle).
- The IR Spectroscopy Principle describes an increase in the energy of atoms rotating around their links.
A molecule’s total energy is the sum of its electronic, vibrational, and rotational energies, all of which are quantized, meaning they have definite energies that are specific to the molecular species.
The relative energy necessary to produce electronic, vibrational, and rotational transitions are as follows:.
Electronic >>> vibrational >> rotational
At room temperature, most of the molecules’ energy is in their ground state. When polychromatic light, or light with a suitable wavelength range, interacts with many identical molecules, the molecules collectively absorb a portion of the light. As a result, certain electrons inside the molecules become stimulated, resulting in an increase in the molecules’ vibrational and rotational energies. The stimulated molecules gained energy in proportion to the increase in electronic energy, as well as a wide range of possible vibrational and rotational transitions,.
ΔE = (Ee + Ev + Er)upper – (Ee + Ev + Er)lower
It is observed that the variation in energy gain follows a Gaussian distribution, with one transition being provided by more molecules than any other and the related transitions provided by fewer molecules. The wavelength of absorbed light, which corresponds to the total energy gained at each transition, is given as


Ultra-Violet spectroscopy
Introduction
UV-visible spectroscopy is absorption spectroscopy in the 100-800 nm wavelength range. In this case, the atoms or molecules experience an electronic transition. The table below illustrates the UV-visible range.

Applications
- It is used to determine the molar composition of complexes.
- It is used for determination of instability constants.
- Used for the determination of pK value of an indicator.
- Used to determine the structure of organic compounds for e.g. lactim-lactam
- Used to determine the structure of inorganic compounds.
- Used to determine the structure of inorganic complexes.
- Used in quantitative analysis.
- Used for determination of molecular weights.
Types of electron present in organic compounds
UV spectroscopy employs the absorption spectroscopy technique. UV absorption spectra result from the transition of an electron or electrons within the molecule, which is triggered by valence electron transitions. Organic compounds have three different sorts of electrons: sigma, pi, and non-bonded. The characteristics of each type of electron are as follows:
Sigma (σ) electrons form saturated bonds like carbon-hydrogen or carbon-carbon.
Pi (Π) refers to electrons found in unsaturated hydrocarbons. Compounds like aromatics and trienes contain electrons.
Non-bonded (n) electrons are those that do not form atom-to-atom bonds in molecules.
The energy required to excite these electrons is distributed as follows:
σ >>>> Π >> n.
UV light has insufficient energy to excite sigma electrons but can excite β and n electrons.
Bonded electrons reside in the ground state, but when exposed to UV radiation, they move to a higher electronic shell. These electrons are anti-bonding and are represented by * in higher electronic shells since they are not in a bonding state.
Transition types in organic molecules
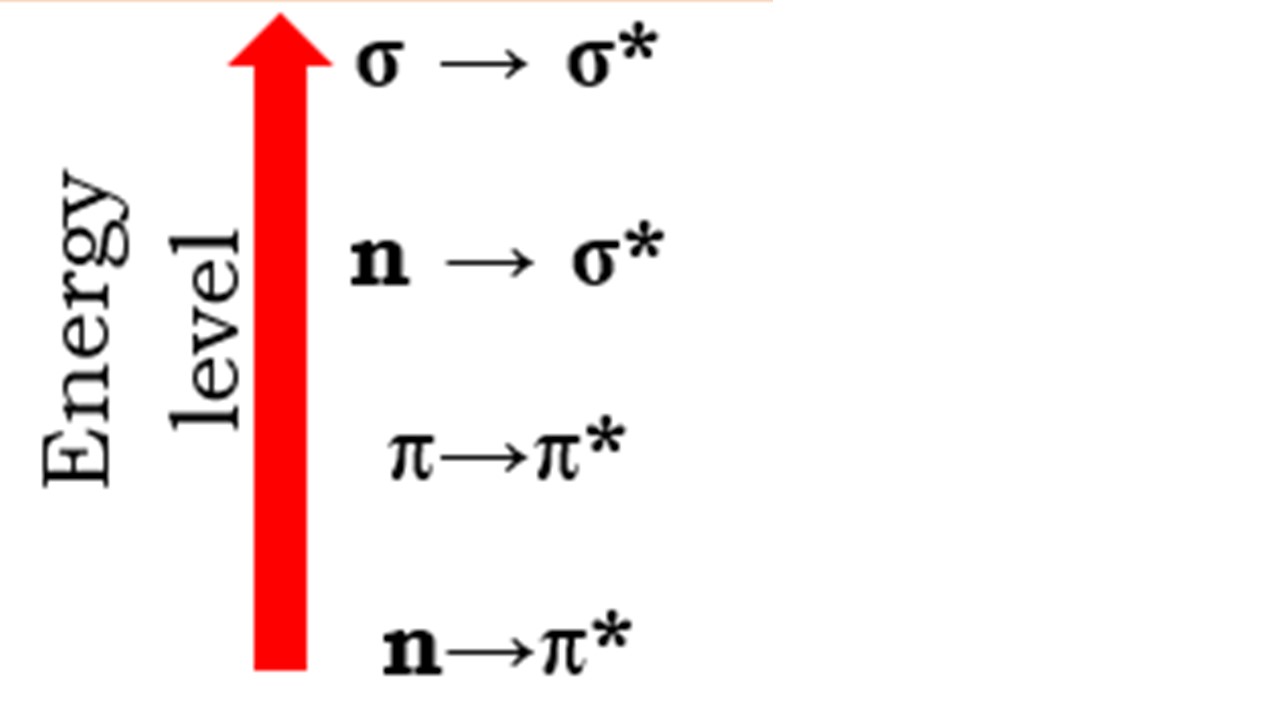
UV radiation absorbed by complex organic compounds causes an electrical change of the valence electrons. Organic compounds typically undergo transitions such as σ →σ*, π →π*, n →σ*, and n →π*. The first three transitions are labelled as allowed transitions, but the last transition, n →π*, is designated a forbidden transition. The specifics of each transition are as follows:
1. σ →σ*
This transition can be caused by covalent bond electrons, which occur in saturated hydrocarbons. In such compounds, there are no lone pairs of electrons since all the electrons are engaged in a single bond. This transition demands a substantial quantity of energy. This transition can be caused by UV light from the far ultraviolet spectrum.
2. π →π*
The π→π* transition occurs when an electron moves from a bonding π orbital to an anti-bonding π* orbital. This transition happens in every molecule with a π bond system (C=C or C≡C). This transition requires less energy than σ→σ* and n→σ*, but more than n→π*. The UV photons have enough energy to cause this transformation.
3. n →σ*
Saturated compounds with lone pair (non-bonding) electrons undergo the n→σ* transition. Compounds like alkyl halides and primary aliphatic amines also undergo the n→σ* transition. A n→σ* transition typically requires less energy than a σ→σ* transition, but more than π→π* and n→π*. The absorption bands appear at longer wavelengths in the vacuum ultraviolet region (180-210 nm)
4. n →π*
This type of transition is shown by unsaturated molecules, which contain atoms such as oxygen, nitrogen, and sulphur. These transitions exhibit a weak band in their absorption spectrum. Compounds like aldehyde, ketones, etc. shows this type of transition. The band due to the n →π* transition generally occurs in the range 270–300 nm and is the least energy transition.
In terms of energy, the above transitions are rated as follows:
Transition probability
It is not necessary for a molecule to undergo an electronic transition when exposed to UV light. The likelihood of an electronic transition is determined by the molar extinction coefficient (λmax) and other factors. The transition likelihood has been separated into two categories: allowed and forbidden.
Allowed transition
Transitions with Emax values greater than or equal to 104 are considered allowed. This form of transition is typically encountered with π→π*.
Forbidden transitions
Transitions with an Emax value less than 104 are considered forbidden. This type of transition is typically encountered with n→π*.
Laws of spectrophotometry and colorimetric
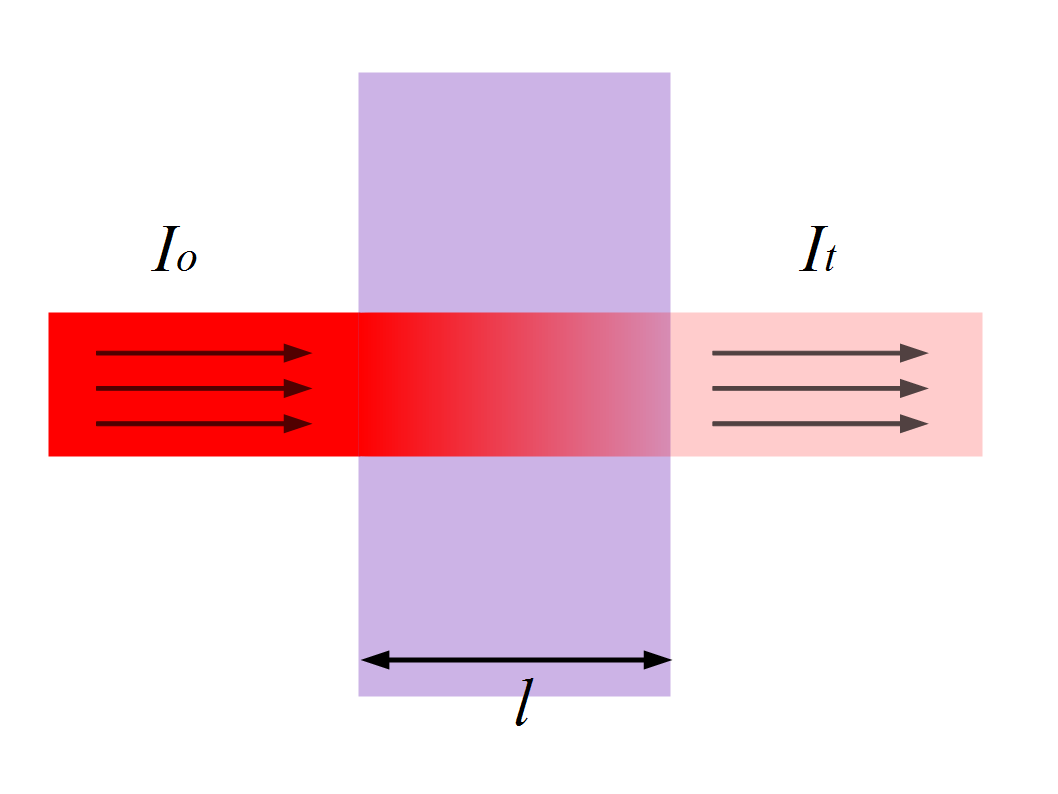
Lambert’s law
Statement
When a light beam is permitted to travel through a clear material, the rate of decrease in intensity with the thickness of the medium is directly proportional to the intensity of the light, and it is stated mathematically as
It = I0e–Kt
Discussion
The equation is obtained by the solving the mathematical expression as –
Change in light intensity with thickness∝Intensity of light

Where,
I = Intensity of light
dI = Change in light intensity
dt = Change in thickness
dI/dt – change in light intensity with the thickness of the medium
Removing the proportionality sign
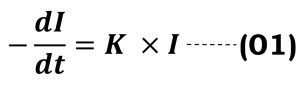
K = proportionality factor
Multiplying the equation (01) by -1, we get-

Rearranging the equation (02), we get-

Now, when there was no thickness, light intensity was “I0”but when thickness “t” is present, light intensity will be “It”. Integrating the equation (03) we get-



By solving equation (05), we get-
InIt ─ InI0 = ─K (t─0)
∴InIt – InI0 = ─ K × t (06)
Multiplying the equation (06) by ─1 we get-
─ InIt + InI0 = K× t (07)
Rearranging equation (07) we get-
InI0 – InIt = K × t (08)
Converting the equation (08) to exponential form, we get-
It = I0e–Kt
Where,
I0: Intensity of the incident light,
It: Intensity of transmitted light,
K: constant,
t: Thickness of the medium
Deviations from Lambert’s law
Lambert’s law, which is based on thickness, showed no variances. The presence of a fingerprint and a rough surface are the sole explanations for a non-linear connection in Lambert’s law.
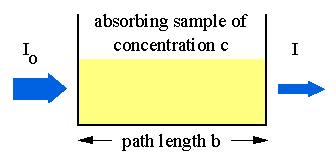
Beer’s Law
When a beam of light passes through a transparent medium, the rate of reduction in intensity with the concentration of solution is exactly proportional to the intensity of the light and is described mathematically as
It = I0e–KC
The mathematical derivation for beer’s law is same as that of beer’s law, the only change is use of concentration “C” instead of thickness “t”
Were,
I0: Intensity of the incident light;
It: is the intensity of transmitted light,
K: constant;
C: Concentration of absorbing substance
Deviation from Beer’s law
According to Beer’s rule, absorbance is proportional to concentration. Thus, when the graph of absorbance vs. concentration is drawn, it produces a straight line that passes through the origin. Deviations are what occur when there is no linear response. There are two sorts of deviations: positive deviations if the curve is above the linear part, and negative deviations if the curve is below it. According to the facts, deviations from Beer’s law arise due to the following factors:
- Beer’s law is not applicable to concentrated solutions.
In concentrated solution, the excited molecules may undergo quenching phenomenon that leads to deviation from linearity.
2. Beer’s law holds a linear relationship over a wide range of concentrations, provided that the chemical entity of the solute remains the same.
3. Beer’s law is not applicable for the dissociating, associating or ionic form of solute. For example, benzyl alcohol in chloroform exists in polymeric form and is written as
4. Deviations from Beer’s law can also occur due to presence of impurities that are also absorbed within the absorption spectrum of solute
5. Deviation also occurs if monochromatic light is not used
6. Deviation may occur if the slit width is not proper. Due to this, stray radiations can reach the detector
7. Beer’s law is not applicable for suspension and emulsions
Beer’s – Lambert’s law
When a beam of monochromatic light is delivered through a solution of absorbing substance, the absorbance of light is directly proportional to the thickness of the medium and the concentration of solution. This is stated mathematically as
A = a × t × c
Where,
A: Absorbance,
a: absorptivity,
t: Path length,
c: concentration of absorbing substance
Some important definitions
Absorption coefficient
It is defined as the reciprocal of the thickness which is required to reduce the light to 1/10 i.e. 0.1 of its original intensity and written mathematically as –
It/I0 = 0.1
Transmittance
The ratio of intensity of light to the intensity of light transmitted is called as transmittance and is written as-

Where,
T is transmittance,
I0 is Intensity of incidence light,
It = Intensity of transmitted light.
Absorbance
Absorbance is defined as the log ratio of intensity of transmitted light to intensity of incident light and is written as

Where,
A is Absorbance,
I0 is Intensity of incidence light,
It = Intensity of transmitted light.
Specific absorbance (E or A)
Specific absorbance is defined as the absorbance of substance whose concentration is expressed in terms of percentage. Mathematically it is expressed as

Molar absorption coefficient or Molar absorptivity (ε)
Molar absorptivity is defined as the absorbance of substance whose concentration is expressed in terms of moles per litre. Mathematically it is expressed as
Chromophore
The term chromophore is defined as any group which exhibits absorption of electromagnetic raditions in the visible or ultraviolet region with or without imparting colour to the solution. e.g. carbonyl, acids, esters, nitrile etc. functional groups. Two types of chromophore are known-
Chromophore with π electrons only
This type of chromophore contains bonds and shows →*. For e.g. acetylene, ethylenes etc functional group
Chromophore with π and n electrons
This type of chromophore contains bonds and functional groups that have non-bonded electrons and shows →* and n→*. For e.g. carbonyl, nitriles, azo etc functional group
Auxochrome
Auxochrome are defined as those functional groups which are not the chromophore but when added to chromophore it shifts the absorption maximum towards longer wavelength along an increase in absorption. For e.g. hydroxyl, amino, ethers etc.
Shifting of λmax and intensity of absorbance
Addition of chromophore and or auxochrome results in alteration of wavelength or intensity of absorbance. The alteration is also seen with change in mobile phase. The different changes in intensity and absorption takes place are-

Bathochromic or red shift (UV → Visible)
Bathochromic or red shift is defined as shifting of absorption maxima (λmax) towards longer wavelength (Visible). This shifting occurs due to addition of auxochrome like hydroxyl (-OH) or amino (-NH2) functional group or because of change in solvent from polar to non-polar.
Hypsochromic shift or blue shift (UV → far UV)
Hypsochromic or blue shift is defined as shifting of absorption maximum (λmax) towards shortest wavelength (Vacuum UV). This shifting of wavelength is observed due to removal of chromophore or change in solvent polarity.
Hyperchromic effect (↑)
Hyperchromic effect is defined as increase in the intensity of absorption. This occurs due to addition of auxochrome.
Hypochromic effect (↓)
Hypochromic effect is defined as decrease in the intensity of absorption. This occurs due to addition of functional group that has capability to distort the geometry of molecules.
Instrumentation
Block diagram of UV

Radiation source
Radiation source is that component of UV spectrophotometer which emits UV radiation. The different lamps like tungsten filament lamp, hydrogen discharge lamp, Deuterium lamp, xenon discharge lamp, mercury arc lamp are used to serve this purpose. From these lists, tungsten filament lamp is used in visible region.
These entire lamps, works on a principal of excitation. The lamps are heated by electric energy releasing electrons. These liberated electrons colloids with the neutral gas molecules that results in electronic, vibrational and rotational excitation. Base on the pressure of gas spectra are emitted. At low pressure line spectra are emitted but at high pressure band and continuous spectra are observed.
The ideal requirement of radiation source is-
- It must be stable
- It must have sufficient intensity that can be detected at the end of optical path
- It must supply continuous radiation over the entire wavelength region.
The details of each lamp are tabulated in table
Sr. No | Name of lamp | Gas/Range | Advantage | Disadvantage |
1 | Tungsten Lamp | – | Lamp is Stable, robust and easy to use for visible region | Maximum radiation is emitted at Near IR range |
2 | Hydrogen discharge | Hydrogen gas 120nm – 350nm | Lamp is Stable, robust and easy to use | —– |
3 | Deuterium discharge | Deuterium gas 120nm – 350nm | Lamp is Stable, robust and easy to use Use specially when high intensity is required | More expensive than hydrogen discharge lamp |
4 | Xenon discharge | Xenon gas | Intensity is more than the hydrogen discharge lamp | ——– |
5 | Mercury arc | Mercury vapour | Used as a standard source in most of UV and produces sharp lines | It is not suitable for continuous spectra studies. |
Monochromator
A Monochromator is an optical device that transmits a mechanically selectable narrow band of wavelengths of light or other radiation chosen from a wider range of wavelengths available at the input. The essential component of monochromator is an entrance slit; dispersion system and exit slit.
The radiation source is use to emits continuous spectra within the UV range. Thus to narrow the wavelength band, monochromator are used. Filters, Prism and or gratings are used as monochromator in UV-Visible spectrophotometer. From this, filters are used in visible spectrophotometer whereas prism and gratings are used in UV spectrophotometer with or without filters.
Filters
A light filter is a device that allows light of the required wavelength to pass but absorbs light of other wavelengths wholly or partially. Thus, a suitable filter can select a desired wavelength band. Two types of filers are available to achieve the desired task viz., absorption filter and interference filter.
Absorption filters
The absorption filters are those filters that selective absorb the unwanted radiations. It is made of solid sheet of glass that has been coloured by a pigment.
Absorption filters are classified in to two types cut off filters or bandpass filters.
Interference filters
Interference filters are those filters that functions on interference phenomenon at desired wavelength allowed the selective wavelength to pass but permits rejection of unwanted radiations by selective reflection. The interference filters are prepared by taking two glass plates. On this plate semitransparent metal film is coated. This metal film is further coated with dielectric material like Magnesium fluoride (MgF2). Another coat of metal film is given on this dielectric material. The last step is put a glass plate on it to provide mechanical stability.
When the ray of light is incident upon an interference filter, a part of light reflects back whereas the remaining light is transmitted. The reflected light is reflected repeatedly by the metal layer. This continues till the desired wavelength is constructed. The interference filers have a band pass of 0.1-0.15nm and the peak transmittance of 40-60%.
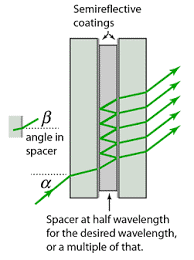
Prism
When the beam of monochromatic light is passes through a prism, the light either get bent or reflected. The amount of this deviation depends on wavelength of monochromatic light used. Based on the use of prism in spectroscopy it is made of different materials. For visible spectroscopy, the prism is made of glass but in case of UV spectroscopy it is made of quartz. Glass causes absorption of UV radiations which makes its incompatible.

Gratings
The function of grating is to disperse the heterochromatic radiation into its component wavelengths. A grating consists of a large number of parallel lines on a highly polished surface such as alumina. These parallel lines are called as grooves. When light rays strike on the grating, the grooves act as scattering centre for light rays. Thus the light is diffracted or spread out over a range of angles as shown in the figure.
Generally, grating is difficult to be prepared. Therefore replica grating are prepared from an original grating. This is done by coating the original grating with the film of an epoxy resin which after setting is removed to yield replica then replica is made reflective by aluminizing its surface.

Slits
There are two slits viz., entrance slit and exit slit. The main function of entrance slit is to provide the narrow source of light so that there should be no overlapping of monochromatic images. Exit slit selects the narrow band of dispersed spectrum for observation by the detector. Generally, entrance and exit slits have equal width.
Cells
The cell holding the sample (generally in solution form) should be transparent to the wavelength region that has being recorded. Cells made of glass are used for visible region where cells made of quartz are used for UV region. The thickness of the cell is generally 1cm (internal distance) but 5 cm and 10 cm cells are also available. Cells may be rectangular or cylindrical with flat ends in shape. The choice of sample cell is based upon
- The transmission characteristics at the desired wavelength
- The path length, shape and size
- The relative expenses
Detection of radiation (detectors)
To detection the UV radiation, three types of instruments are used. The basic principle lies in all these instruments are measurement of electrons by photosensitive devices. Three types of photosensitive devices are used namely photovoltaic cell also called as barrier layer cell, photo tubes and photo multiplier tubes.
Photovoltaic cell (Barrier layer cell)
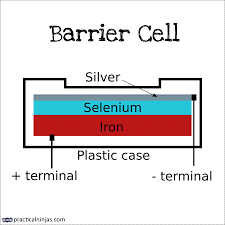
This cell is entirely different in design and principle from the other photo emissive cell. Barrier layer cells operate without the use of battery.
Construction
Barrier layer cell consist of two electrodes. The first electrode is made of metal base plate like iron or aluminium. On the metal surface a thin layer of semi-conductor metal like selenium is deposited. On this layer of semi-conductor, a very thin layer of silver or gold is applying which acts as a second electrode.
Working
When the radiation is fall on the surface of selenium, electrons are generated at the selenium-silver surface. These electrons are collected by the silver. The accumulation of electrons on the silver surface creates an electric voltage difference between the silver surface and metal electrode. Due to this photocurrent will flow that is directly proportional to intensity of incident radiation beam. If this cell is connected to a galvanometer, current will flow which will varies with the intensity of incident light.
Advantage
Barrier layer cell offers the advantages of being rugged and requires no external power supply.
Disadvantages
- Barrier layer cells are generally use for visible region and not for UV region.
- From the whole visible region, these cells are less sensitive in blue region than in the green and yellow region.
- The current output of photovoltaic cells depends upon the wavelength of the incident light.
- The current produce by photovoltaic cell cannot be readily amplified by conventional electronic circuits because of the low internal resistance.
- In general, photovoltaic cells show fatigue effect. In this cells the initial photocurrent is very high and its attends the steady value after a few minutes.
Phototubes
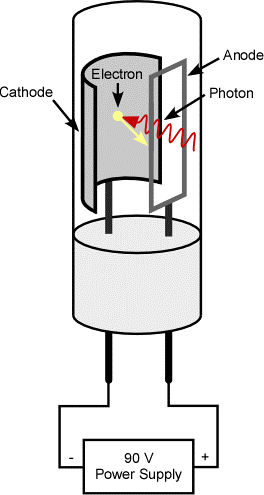
These cells are also known as photo-emissive cells. The choice of sample cell is based upon.
- The transmission characteristics at the desired wavelength.
- The path length, shape, and size of cell
- The relative response
Construction
Phototubes consist of an evacuated glass blub. Inside the bulb there is a light sensitive cathode in the form of half cylinder of metal. The inner surface of cathode is coated with a light sensitive layer such as caesium or potassium oxide and silver oxide. The metal ring inserted near the centre of bulb acts as an anode.
Working
When the radiation is fall on cathode photoelectrons are emitted. This are attracted towards anode. The electrons collected at anode are returned via the external circuit. Flowing of this electron leads to generation of current that is proportional to amount of light striking at photosensitive surface.
Advantages
Phototubes are more sensitive than barrier layer cell. Phototubes have high internal resistance their output current can be easily amplified.
Photomultiplier tubes
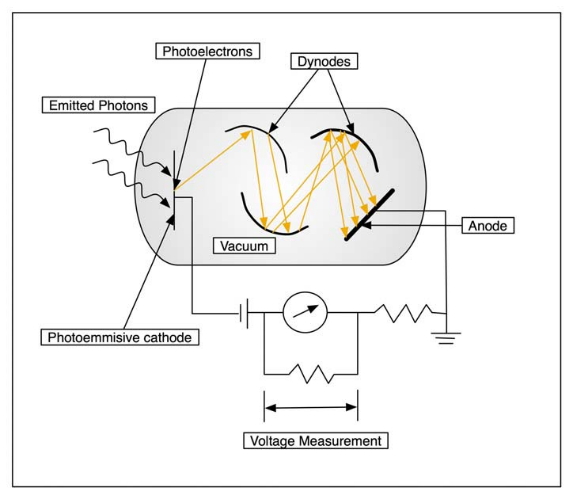
The sensitivity of photo-emissive cells can be increased considerably by employing photomultiplier tube.
Construction
The photomultiplier tube consists of an electrode covered with photo-emissive material. This tube also contains large number of plates known as dynodes. Each dynode is covered with a material that emits several electrons for each electron striking on its surface.
Working
When the radiation light is incident upon the cathode surface, electrons are emitted. These liberated electrons are accelerated towards sensitive surface of the dynode where secondary electrons are emitted in greater number than initially striking the plate. These electrons in turn are accelerated towards another surface of next dynode which was maintain at higher potential where the number of electrons is again increased by a factor of four or five. The process is repeated several times until large numbers of electrons arise at the collector. The number of electrons falling on the collector measures the intensity of light incident on the cathode surface. Most photomultiplier tubes have about 10 dynodes. Each dynodes is maintain at 75-100V more positive than the previous dynode.
Photomultiplier tubes
Working
When the radiation light is incident upon the cathode surface, electrons are emitted. These liberated electrons are accelerated towards sensitive surface of the dynode where secondary electrons are emitted in greater number than initially striking the plate. These electrons in turn are accelerated towards another surface of next dynode which was maintain at higher potential where the number of electrons is again increased by a factor of four or five. The process is repeated several times until large numbers of electrons arise at the collector. The number of electrons falling on the collector measures the intensity of light incident on the cathode surface. Most photomultiplier tubes have about 10 dynodes. Each dynodes is maintain at 75-100V more positive than the previous dynode.
Single beam and double UV Spectrophotometer


Quantitative estimation of substance
For single component
The quantitative estimation of absorbing substance can be quickly carried out by preparing a solution in a transparent solvent and measuring its absorbance at a suitable wavelength. Generally, the maximum absorbance wavelength is used for the estimation. While measuring the absorbance of the analyte it is desirable that absorbance given by the analyte should not be additive absorbance. To convert the absorbance into concentration different methods are used. The details of each method are as follows-
1. Calibration graph method
In this method, the number of standard solutions were prepared using reference substance and their absorbance were recorded. After getting the absorbance the graph was plotted by taking concentration on x-axis and absorbance on y-axis. The linear graph was then constructed passing from origin. Now, measure the absorbance of test analyte solution and put on the linear curve and from that determine the concentration of unknown solution.
2. Linear equation method
In this method first the linear curve was constructed. After constructing the linear curve, the equation for straight line was obtain i.e. Y = mx + C.
Where,
Y → Absorbance of unkwown solution,
m → slope of line,
x → concentration of unknown solution,
C → Y or X intercept.
Putting the value of absorbance of unknown solution in this equation, directly the concentration of test solution can be determined.
3. By using standard absorptivity value
This method was widely used by various pharmacopeia. This method is applicable for the stable analyte that are remains unaffected by variation in instrumental parameters like scanning speed, slit width etc. The use of standard absorptivity values prevents the preparation of standard solution of the reference standard substance for determination of absorptivity. It is also advantageous where the reference standard is difficult to obtain, or it is more expensive. The concentration of analyte is calculated by using following formula.
A = ε x c x b
Where,
A → Absorbance of test analyte,
ε → Molar absorptivity,
c → Concentration of test analyte,
b → cell path length
4. Using single point standardization method
Single point standardization method involves the measurement of the absorbance of a sample solution and standard solution. The sample solution and standard solution are prepared in identical manner. The absorbance of standard and test solution is determined. From the list of standard absorbance, one absorbance is selected which has absorbance close to the absorbance of test solution. Now the concentration of test solution is determined by using following formula

Where,
Ctest → Concentration of test solution,
Cstd → Concentration of standard solution,
Atest → Absorbance of test solution,
Astd → Absorbance of standard solution.
5. By using double point standardization method
In the single point standard, accuracy is limited. To overcome this problem multiple point standardization method is used. In the method series of standard solution were prepared. In the identical manner the test solution was also prepared. The absorbance of all solution was recorded. From the absorbance of standard solutions, two absorbance were selected. The selection was done with respect to the test solution absorbance. The first standard absorbance that was selected has the absorbance less than test absorbance. The second standard absorbance which was selected has the absorbance more than standard solution. The concentration of test solution was determined from the formula as

Where,
Ct → concentration of test solution,
At → Absorbance of test solution,
Cs1 and Cs2 → concentration of standard1 and standard2 solution (1>2) respectively
As1 and As2 → Absorbance of standard1 and standard2 solution respectively
For Multiple components analysis
When two or more analyte are present in the solution and each have independent absorbance that do not interfere in one another absorbance then the individual concentration can be determined by one of the above method. But if the absorbance is interfered then the concentration of analyte can be determined from any of the above methods. In this case concentration of individual analyte can be determined by using
1. Extraction method
In this method, the mixtures of analyte are mixed with suitable solvent. The analyte which is more affinity for the solvent will get dissolve whereas the analyte that has low or poor affinity for the given solvent will remain in precipitate form. Thus by doing simple filtration, two different analyte get separated and can be individual identified using any one method of single component analysis.
2. Simultaneous equation method (Vierodt’s method)

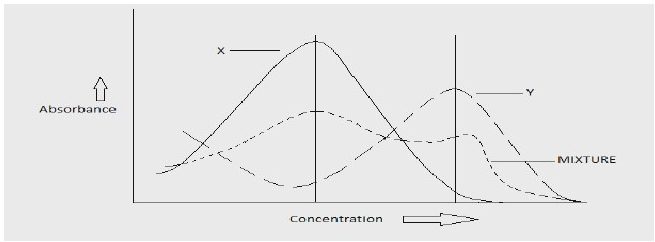
In this method, the mixtures of analyte are not separated but assay simultaneously hence called as simultaneous equation method. Consider the two analyte “X” and “Y” which has its own lmax.
The information that required is
- The absorptivity’s of X at λ1 and λ2 are “ax1” and “ax2” respectively
- The absorptivity’s of Y at λ1 and λ2 are “ay1” and “ay2” respectively
- The absorbance of sample at λ1 and λ2 are “A1” and “A2” respectively
- The concentration of X and Y in dilute sample are Cx and Cy respectively
Now using the equation from Lambert’s-Beer’s law, we get
A = acb
Two equations are constructed based upon the fact that at λ1 and λ2 the absorbance of the mixture is the sum of the individual absorbances of X and Y.
Thus at λ1
A1 = ax1 ×b × cx + ay1 × b × cy
At λ2
A2 = ax2 ×b × cx + ay2 × b × cy
Since the measurement is generally done in 1cm cell, so putting the value of b as 1, we get
A1 = ax1 × cx + ay1 × cy (01)
A2 = ax2 × cx + ay2 × cy (02)
Rearranging the equation (01), we get
A1 – ay1 × cy = ax1 × cx

Rearranging the equation (02), we get
A2 – ax2 × cx = ay2 × cy

Putting value of Cy from equation (04) in equation (03), we get


To obtain the maximum precision, the absorbance ration as given below should lie outside the range of 0.1-2.0.

These criteria will be satisfied only when the λmax of two analyte are reasonably dissimilar.
3. Q- Absorbance method/Absorbance ratio method

The absorbance ratio method is a modification of simultaneous equations procedure. It is based on the fact that the analyte obeys Beer’s law at all wavelengths and the ratio of absorbance at any two wavelengths is a constant value which is independent on concentration. The ratio of absorbance is defined as Q value and thus the name given as Q –Absorbance method.
Thus in absorbance ratio method, absorbance are measured at two wavelengths one being the λmax of any one component and the other being a wavelength of equal absorptivity of two components (λ1 as shown in figure above). The λ1 is called as iso-absorptive point.
Derivative spectroscopy
Derivative UV spectroscopy is a powerful analytical technique that extends ultraviolet absorption spectroscopy to reveal even finer details about molecular interactions and structural properties. Derivative UV spectroscopy, like a magnifying glass, brings hidden patterns into focus, making it an essential tool in sectors ranging from medicines to environmental monitoring.
The absorption of light by a sample across a variety of wavelengths is measured in traditional UV spectroscopy (zero order derivative). The obtained spectrum provides information about the overall absorbance behaviour, but it can occasionally mask the presence of narrow features and transitions. This is when derivative UV spectroscopy comes into play.
The mathematical differentiation of the absorption spectra is involved in derivative spectroscopy. This converts the absorbance values into the rate of change of absorbance with respect to wavelength, effectively emphasizing even minor differences in the spectrum. Peaks, dips, and inflection points that were previously buried in the original spectrum become considerably more visible in the derivative spectrum.
Obtaining the derivative orders spectrum

Derivative spectroscopy converts normal or zero order spectrums to their first, second, or higher derivative spectrums. It results in significant changes in the form of the derivative obtained. An appropriate choice of derivative order allows for meaningful separation of overlapping signals. The best derivative order achieves criterion such signal height, width, and distance between maxima in basic spectrum, with low orders predicted for large spectrum bands and higher orders expected for narrow spectral bands. A Gaussian band depicts an ideal absorption band and provides a clear picture of the transition that occurs in the derivative spectra. When absorbance is plotted against wavelength, a graph is produced that shows a peak with maxima and minima (also points of inflection) that is meant to pass through zero on the ordinate.
Zero order derivative spectroscopy
The zero order derivative is the first step in producing more derivatives. In other words, a zeroth order spectrum can yield an nth order derivative. The D0 spectrum, or zeroth order, is a characteristic component of the normal absorption spectrum in derivative spectroscopy.
If a spectrum is represented in terms of absorbance (A) as a function of wavelength (λ), the derivative spectra is provided as A= f (λ).
First order derivative spectroscopy
“First order” or “first derivative” in spectroscopy refers to the mathematical procedure of computing the rate of change of a function with respect to a wavelength (λ). The primary idea underlying first order derivative spectroscopy is to highlight the rates of change in absorbance of the features and transitions contained in an absorption spectrum.
The absorbance spectrum obtained in UV-Vis spectroscopy is a plot of how much light a sample absorbs at different wavelengths. It shows the relationship between absorbance and wavelength and often includes peaks that correspond to specific transitions or components within the sample.
The first order derivative spectrum is a plot of absorbance change with wavelength versus wavelength, i.e. the rate of absorbance change with wavelength,
dA/dλ = f'(λ)
Even in derivatized form, the spectrum is more complicated than the zero-order spectrum. As the absorbance band’s maximum, first order spectra pass through zero. The absorbance band of the first order derivative has positive and negative peaks and minima.
Construction of first order derivative spectrum
- Original Absorbance Spectrum (A): This is the initial UV-Vis absorption spectrum obtained for the sample, showing absorbance values (A) as a function of wavelength (λ).
- Calculation of First Derivative (ΔA/Δλ): The first derivative of the absorbance spectrum is calculated by determining the rate of change of absorbance (ΔA) with respect to the change in wavelength (Δλ). Mathematically, this is expressed as dA/dλ, where dA is the change in absorbance and dλ is the change in wavelength. In numerical terms, the first derivative can be approximated as the difference in absorbance values at neighbouring wavelengths.
- Plotting the First Derivative Spectrum: The resulting first derivative spectrum is plotted, where the y-axis represents the rate of change of absorbance (dA/dλ), and the x-axis remains the original wavelength values. The peaks and troughs in the first derivative spectrum correspond to points of significant change in absorbance in the original spectrum.
Advantages of First Order Derivative Spectroscopy
- Peak Resolution: Overlapping peaks in the original spectrum become separate, distinct peaks in the first derivative spectrum, aiding in peak identification and quantification.
- Background Correction: Baseline shifts and background interference are minimized, improving accuracy in quantitative analysis.
- Sensitivity Enhancement: First order derivative spectroscopy enhances sensitivity, making it useful for detecting trace components or subtle variations in samples.
- Identifying Spectral Features: It allows for a clearer identification of subtle spectral features that might not be evident in the original spectrum.
Second order derivative spectroscopy
Second order derivative spectroscopy, commonly known as the second derivative or Δ^2A spectroscopy, is a spectroscopic analysis technique for enhancing and extracting detailed information from absorption spectra. It is based on first order derivative spectroscopy principles, however rather of calculating the rate of change of absorbance with respect to wavelength (dA/dλ), it calculates the rate of change of the first derivative with respect to wavelength (d2A/dλ2).
The second derivative is exactly proportional to the concentration. The ratio d2A/dλ2 must be significant, the greater the ratio, the greater the sensitivity. The approach may be used to generate atomic and gas molecular spectra.
d2A/dλ2=f”'(λ)
Advantages of Second Order Derivative Spectroscopy
- Enhanced Peak Resolution: Second order derivative spectroscopy greatly improves the resolution of absorption peaks, allowing for the clear identification of closely spaced peaks.
- Clearer Spectral Features: Fine details, subtle transitions, and small shifts in the original spectrum are highlighted in the second derivative spectrum, making it easier to distinguish spectral features.
- Background Correction: Baseline shifts and background interference are minimized, improving accuracy in quantitative analysis.
- Sensitivity Enhancement: Second order derivative spectroscopy enhances sensitivity, making it effective for detecting trace components and subtle spectral changes.
- Structural Insights: It can provide valuable insights into molecular interactions, structural characteristics, and the composition of the sample.
Third order derivative spectroscopy
Third order derivative spectroscopy, commonly known as Δ^3A spectroscopy, is a sophisticated derivative spectroscopy method that is utilized in analytical chemistry and spectroscopy. It extends the ideas of first and second order derivative spectroscopy to improve the spectral characteristics and details in an absorption spectrum. The third derivative of the absorbance spectrum with respect to wavelength (λ) is determined in third order derivative spectroscopy.
Third order derivative spectroscopy is an effective technique for researchers and analysts that need exceptionally high spectral resolution and the capacity to reveal minute details in UV-Vis absorption spectra. It is used in industries like as pharmaceuticals, environmental studies, and materials research, where exact component identification and quantification are crucial.
d3A/dλ3= f”'( λ )
Advantages of Third Order Derivative Spectroscopy
- Ultra-High Resolution: Third order derivative spectroscopy provides an even higher level of spectral resolution compared to first and second order derivative spectroscopy. It is exceptionally useful for differentiating closely spaced peaks and identifying subtle spectral features.
- Enhanced Detail: Fine spectral details, small shifts, and minor transitions that may be obscured in lower-order derivative spectra become much more pronounced in the third derivative spectrum.
- Structural Insights: It can provide valuable insights into molecular interactions, structural characteristics, and the composition of complex samples.
- Background Correction: Like other derivative spectroscopy techniques, third order derivative spectroscopy minimizes baseline shifts and background interference, improving the accuracy of quantitative analysis.
- Sensitivity Enhancement: Third order derivative spectroscopy enhances sensitivity, making it effective for detecting trace components and subtle spectral changes.
Fourth order derivative spectroscopy
Fourth order derivative spectroscopy, commonly known as Δ^4A spectroscopy, is a method used in analytical chemistry and spectroscopy. It tries to increase spectral characteristics and extract fine details from an absorption spectrum, like lower-order derivative spectroscopies (first, second, and third order). The fourth derivative of the absorbance spectrum with respect to wavelength (λ) is determined in fourth order derivative spectroscopy.
The fourth derivative (UV-high pressure) preferentially determines narrow bands.
d4A/dλ4= f”(λ)
Advantages of Fourth Order Derivative Spectroscopy
- Unprecedented Resolution: Fourth order derivative spectroscopy provides an exceptionally high level of spectral resolution, surpassing lower-order derivative techniques. It is valuable for differentiating closely spaced peaks and detecting extremely subtle spectral features.
- Enhanced Detail: It can reveal the finest spectral details, such as minor shifts, subtle transitions, and hidden components within a complex absorption spectrum.
- Structural Insights: Fourth order derivative spectroscopy can provide valuable insights into molecular interactions, structural characteristics, and the composition of complex samples.
- Background Correction: Like other derivative spectroscopy techniques, it minimizes baseline shifts and background interference, improving the accuracy of quantitative analysis.
- Sensitivity Enhancement: Fourth order derivative spectroscopy is highly sensitive and is useful for detecting trace components and subtle spectral changes.
Advantage of derivative spectroscopy
Derivative UV spectroscopy offers several advantages:
- Enhanced Sensitivity: Derivative spectroscopy amplifies subtle changes in the absorption spectrum, making it a remarkable tool for detecting impurities or subtle changes in molecular structure.
- Peak Identification: Peaks that overlap in the original spectrum can be more easily distinguished and identified in the derivative spectrum.
- Quantitative Analysis: Derivative spectroscopy simplifies quantitative analysis by eliminating baseline drift and background interference.
- Structural Insight: It helps in elucidating structural characteristics and molecular interactions that might not be apparent in the standard absorption spectrum.
C. Chemical Derivatization
Chemical derivatization is the indirect method of spectroscopic assay. In this method analyte is chemically modified with the help of suitable excess reagent that produces a new compound which have different spectral properties. Chemical derivatisation methods may be adopted for any of the reasons given below
- For those analytes that do not or poorly absorbs in the UV region. For such analyte, a more sensitive method of assay is obtained by converting the substance to a derivative with a more intensely absorbing chromophore. For e.g. the absorption of sugars in the UV region is very poor. Its quantitative estimation can be done by treating the sugars with anthrone reagents in presence of concentrated sulphuric acid.
- If the quantitative estimation of analyte cannot be done due to presence of interfering components. For e.g. reactions of ketosteriods with hydrazide reagents produces a compound that shows absorption in visible region. The absorption in this region is free from interfering substance.
- Such methods are also used for indirect spectrophotometric analysis where the absorption of analyte is strongly interfered with other UV absorbing substance. For e.g the analysis of adrenaline in procaine injection is strongly interfere with procaine and bactericide agent chlorocresol. Ferrous ions selective reacts with adrenaline that forms violet-coloured compounds.
- Such methods are preferred if cost of estimation is prime consideration. The cost of colourimeter is quite low than the UV spectrophotometer.
The methods that are well documents for converting the analyte in to another compound that shows absorption in the visible region are
Diazotisation method for aromatic amines
Diazotisation is the process where the primary aromatic amines are converted to diazo compounds with the help of nitrous acid in presence of mineral acid like HCl. The diazo compounds are highly unstable and very reactive. The stability of diazp compounds is increased by coupling with suitable coupling reagent. The most widely used coupling reagent are -napthol, β-napthol, N-(1-naphthyl)-ethane-1,2-diammonium dichloride (The bratton-marshall reagent). The reaction involved in this is shown below.
Ar–NH2 + HNO2 + H+ → Ar-N+≡N + 2H2O
Ar–N+≡N + Ar′–H → Ar–N=N–Ar′
This method was adopted by british pharmacopeia for detecting amine impurities in frusemide, iothalamic acid and iodipamide meglumine injection.
Condensation reactions
Many functional groups can be directly and efficiently condensed with other functional group at an ordinary condition like condensation of ketone and amines. Based on type of amine used, different condensation reaction can be formed.

The assay of Nandralone decanoate injection, Betamethasone sodium phosphate injection is based on this method. In both assay isoniazid is used as condensation reagents.
In the assay of methyl testosterone tablets, 2,4-dinitrophenyl hydrazine is used as condensation reaction.
Reduction reactions

The α-Ketol steroids like corticosteroids are assay by this method. The triphenyl tetrazolium chloride is used as a reagent which gets reduced to form triphenylformazan the red coloured compound. The concentration of red coloured compound is determined at 485 nm. The reaction involve in this method is written as shown above
Oxidation method
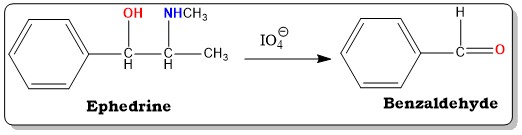
Oxidation of side chain having simple phenyl group can be done with the help of oxidising agent. Due to this the carbonyl group is formed which shows better absorption in the UV region than the parent compound. The widely used oxidising agent is alkaline potassium permanganate solution, acidified potassium dichromate solution or perchlorate solution. The assay of ephedrine is done by using this method and reaction involved in this method is written as
Metal ligand Complexation
Many organic reagents form complexes with metal atoms by forming the coordinate bonds. The chelate forms by multidentate ligands are often coloured and their concentration can be determined by visible spectroscopy. For example, determination of lead by using dithiazone, estimation of adrenaline by using ferrous sulphate solution are done by this method. The coloured develop depends on the pH and changes in pH leads to change in colour formation.




